Dr. Mitra Basu Chhillar, M.D.,M.B.A.,F.A.M.
Functional & Regenerative Medicine Specialist
Medical Director, SOMA Wellness Clinic
Introduction
In an era where chronic fatigue, hormonal imbalance, anxiety, and burnout have become distressingly common, there is an increasing need to revisit powerful yet underutilized tools in functional and regenerative medicine. One such tool is pregnenolone, often called the “mother of all hormones.”
Discovered in the 1930s, pregnenolone is a naturally occurring steroid hormone synthesized primarily in the adrenal glands, brain, and gonads. It serves as a precursor for the synthesis of other crucial hormones such as DHEA, cortisol, progesterone, estrogen, and testosterone. In recent years, interest has resurged around this foundational molecule for its role in adrenal health, cognitive vitality, anti-aging, stress resilience, hormonal rebalancing, and overall wellbeing.
At SOMA Wellness Clinic, we use sublingual pregnenolone drops in a carefully guided protocol that bypasses common limitations of oral administration. This article explores the scientific rationale, clinical indications, method of use, and emerging research supporting the thoughtful use of pregnenolone in today’s overstressed and hormonally depleted population.
Why Sublingual Pregnenolone?
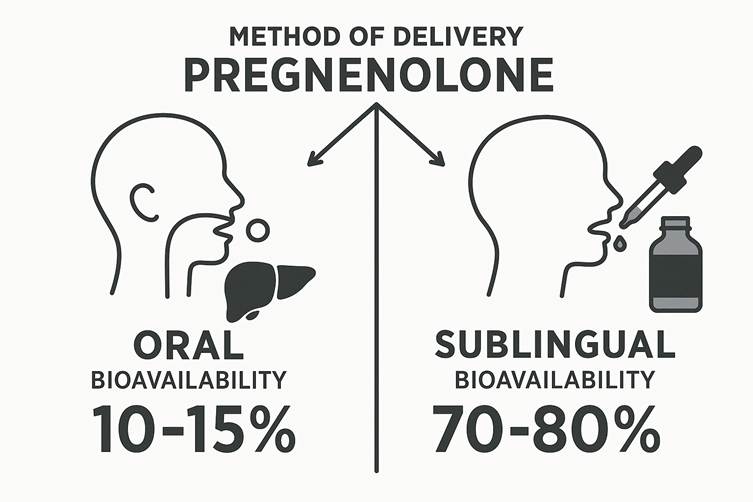
The method of delivery dramatically influences a hormone’s effectiveness. Oral pregnenolone undergoes extensive first-pass metabolism in the liver, significantly reducing its bioavailability. Various studies suggest that oral pregnenolone has a bioavailability of only 10-15%, with most of it being converted to inactive metabolites before reaching systemic circulation.
Sublingual delivery, on the other hand, allows the hormone to bypass the liver, entering the bloodstream directly through the rich network of capillaries under the tongue. This increases bioavailability up to 70-80%, enabling a lower dose to achieve physiological effectiveness.
At SOMA Wellness Clinic, our patients are advised to:
- Shake the bottle well before each use.
- Place 3 to 4 drops under the tongue, early in the morning.
- Avoid using pregnenolone in the evening, as it can increase mental alertness and potentially disturb sleep.
Biochemistry of Pregnenolone
Pregnenolone is synthesized from cholesterol via the mitochondrial enzyme P450scc (CYP11A1). It is the first step in the steroidogenic cascade and gives rise to glucocorticoids, mineralocorticoids, androgens, and estrogens through various enzymatic pathways.
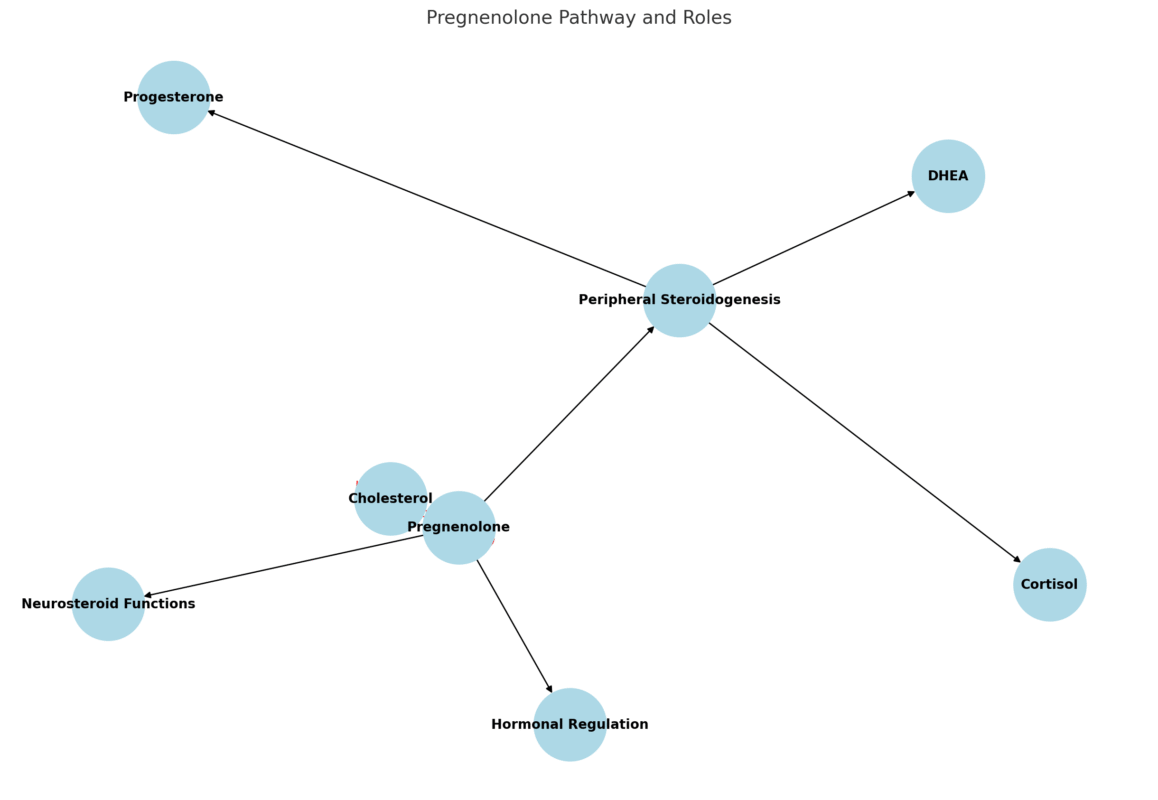
It has multiple roles:
- As a neurosteroid, it directly modulates GABA-A receptors, NMDA receptors, and sigma-1 receptors in the brain.
- In peripheral tissues, it contributes to downstream synthesis of DHEA, progesterone, and cortisol.
- It acts as a regulatory switch that governs the balance between anabolic and catabolic hormone production.
Clinical Indications and Benefits
1. Adrenal Exhaustion and Chronic Stress
Chronic stress overactivates the HPA axis, leading to adrenal dysregulation and downstream hormonal imbalances. In adrenal fatigue, pregnenolone levels often plummet, resulting in reduced DHEA, cortisol, and other steroid hormones.
Supplementing pregnenolone helps recharge adrenal function, restoring the body’s capacity to handle physical and psychological stressors. It also stabilizes mood and reduces anxiety by modulating neurosteroids in the brain.
2. Cognitive Function and Mood
Pregnenolone enhances:
- Memory and learning by modulating NMDA and AMPA receptors.
- Mood stabilization by influencing GABAergic tone and dopamine sensitivity.
Studies show pregnenolone’s potential in supporting patients with depression, bipolar disorder, schizophrenia, and age-related cognitive decline.
3. Anti-aging and Cellular Vitality
Low pregnenolone is associated with:
- Cellular senescence
- Mitochondrial dysfunction
- Reduced hormone synthesis
Supplementation restores cellular resilience, boosts mitochondrial output, and supports neurogenesis. As a result, patients report increased energy, motivation, skin glow, and muscular strength.
4. Hormonal Rebalancing in Men and Women
Pregnenolone is a critical precursor for both estrogen and testosterone. In men and women suffering from low testosterone, estrogen dominance, or progesterone deficiency, pregnenolone restores upstream balance, facilitating endogenous production of downstream hormones.
In women, it helps with:
- Perimenopause and menopause
- Estrogen-progesterone imbalance
- PMS and mood swings
In men, it improves:
- Libido and sexual performance
- Motivation and drive
- Andropause-related fatigue
5. Low Cortisol and Burnout
In individuals with low cortisol levels, pregnenolone offers a safe and physiological method to support the steroidogenic pathway without directly giving exogenous corticosteroids. This is particularly beneficial in:
- Long COVID fatigue
- Post-viral burnout
- Overtraining syndrome
6. Low DHEA-S Levels
DHEA-S is an important marker of vitality and immune resilience. Pregnenolone acts as a parent molecule to DHEA, thus helping restore DHEA-S levels without bypassing upstream regulatory mechanisms.
7. Chronic Fatigue Syndrome and Fibromyalgia
These complex conditions involve neuroinflammation, mitochondrial dysfunction, and HPA axis dysregulation. Pregnenolone’s combined neurosteroid, anti-inflammatory, and mitochondrial-enhancing roles make it an important consideration in their management.
8. Low Libido and Sexual Dysfunction
Sex hormone synthesis, libido, and arousal are deeply influenced by adrenal and gonadal hormone production. Pregnenolone restores foundational endocrine harmony that enhances:
- Desire and performance
- Arousal and stamina
- Emotional connection and energy
Clinical Considerations
- Dose titration should be guided by symptoms and serum hormone levels.
- Periodic monitoring of DHEA-S, cortisol, testosterone, estradiol, and progesterone is recommended.
- Avoid high evening doses, as pregnenolone can increase alertness and interfere with sleep.
Why It’s Often Undiagnosed
Despite its critical role, pregnenolone deficiency is seldom tested in conventional medicine. This is because:
- Labs rarely include pregnenolone in standard hormone panels.
- There is lack of awareness among mainstream practitioners.
- Patients often present with vague symptoms like fatigue, low motivation, anxiety, or insomnia that are misattributed.
Functional and regenerative medicine takes a systems biology view, placing pregnenolone at the center of metabolic, cognitive, and hormonal health.
Contraindications and Cautions
- Pregnant or lactating women should avoid use unless medically supervised.
- Not advised in estrogen-sensitive cancers without guidance.
- May interact with GABAergic medications or mood stabilizers in sensitive individuals.
Scientific Publications and Research Evidence
- Flood JF, et al. Proc Natl Acad Sci. 1992; “Pregnenolone enhances memory in aged mice.”
- Marx CE, et al. Biol Psychiatry. 2009; “Pregnenolone in schizophrenia and schizoaffective disorder.”
- Ritsner MS, et al. Eur Neuropsychopharmacol. 2010; “Pregnenolone as adjunctive therapy for cognitive deficits.”
- Mellon SH, Griffin LD. Brain Res Brain Res Rev. 2002; “Neurosteroids: biochemistry and clinical significance.”
- Genazzani AR, et al. Menopause. 2003; “Neurosteroid role in menopausal syndrome.”
- Goodyer CG, et al. J Clin Endocrinol Metab. 1995; “Age-related changes in steroidogenesis.”
- Vallee M, et al. J Neurosci. 2001; “Pregnenolone modulates anxiety behavior via GABA-A.”
- Maninger N, et al. Psychoneuroendocrinology. 2009; “Neurosteroids and stress resilience.”
- Labrie F. Endocr Rev. 1991; “DHEA and pregnenolone interplay.”
- Pruessner JC, et al. Neuroimage. 2010; “HPA axis regulation and neurosteroids.”
Final Thoughts: Reimagining Resilience
As modern life continues to challenge our biological resilience, pregnenolone offers a science-backed, elegantly simple way to restore the body’s foundational vitality. Whether it’s reversing burnout, balancing hormones, or supporting cognitive performance, pregnenolone is not merely a supplement—it is a strategic intervention in the art and science of regeneration.
For doctors exploring functional protocols, or individuals seeking a deeper solution to chronic fatigue, low libido, and hormonal disarray, sublingual pregnenolone may prove to be a game-changer.
Disclaimer
This article is intended for informational purposes only. It does not constitute medical advice, diagnosis, or treatment. Always consult your physician or qualified health provider before starting any new supplement or therapy. The use of pregnenolone should be medically supervised, particularly in individuals with complex hormonal conditions or psychiatric histories.
Authored by:
Dr. Mitra Basu Chhillar
© 2025, SOMA Wellness Clinic. All rights reserved.
Dr. Mitra Basu Chhillar, M.D., M.B.A., F.A.M.
Medical Director,
SOMA Wellness Clinic
Introduction
Ozone therapy, the controlled administration of medical-grade ozone (O₃), has been explored for over a century, initially for water disinfection and wound treatment, and now for a broad range of medical conditions, including chronic inflammatory diseases, infections, circulatory disorders, and antiaging applications. Despite its historical use, ozone therapy remains controversial due to limited large-scale clinical trials and regulatory skepticism, notably from the U.S. Food and Drug Administration (FDA), which prohibits its medical use citing insufficient evidence of safety and efficacy [1]. However, countries like Germany, Italy, Russia, and Cuba integrate it into healthcare systems under strict guidelines, supported by emerging research on its biochemical mechanisms.
This lecture, designed for medical doctors, provides a detailed examination of ozone therapy’s biochemical basis, mechanisms of action, dosing protocols, clinical applications, and potential in antiaging, with a focus on the biochemical pathways involved. It addresses the user’s request to ensure all 46 references cited in the text are listed, correcting the previous discrepancy where only 25 were provided. By the end, clinicians will have a balanced perspective to evaluate ozone therapy’s role in practice responsibly.
Biochemical Basis of Ozone Therapy
The therapeutic potential of ozone therapy hinges on its ability to induce controlled, mild oxidative stress that activates protective biochemical pathways. Below, we explore the key mechanisms and pathways involved.
Formation of Ozone Peroxides
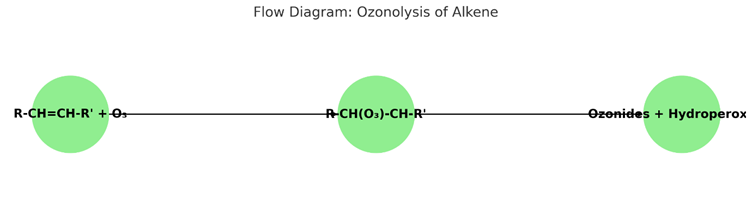
At low doses, ozone selectively reacts with mono-unsaturated fatty acids (e.g., oleic acid) in cell membranes, forming ozonides and hydroperoxides, collectively termed “ozone peroxides.” These intermediates decompose into secondary products like aldehydes, ketones, and hydrogen peroxide (H₂O₂), which act as signaling molecules. Unlike reactions with polyunsaturated fatty acids, which can lead to harmful lipid peroxidation, this selective interaction minimizes cellular damage [2, 3]. The chemical reaction can be represented as:
R-CH=CH-R’ + O₃ → R-CH(O₃)-CH-R’ → Ozonides + Hydroperoxides
Glutathione Interaction
Ozone peroxides are rapidly reduced by glutathione (GSH), the body’s primary antioxidant, generating reactive oxygen species (ROS) and lipid ozonation products (LOPs), such as 4-hydroxynonenal (4-HNE). The GSH/GSSG (glutathione/oxidized glutathione) balance is critical, serving as a limiting factor for safe ozone dosing. Therapeutic doses induce oxidative eustress, promoting cellular protection, while excessive ozone can deplete GSH, leading to oxidative distress [4]. This balance ensures ozone acts as a bioregulator rather than a toxic agent.
Key Biochemical Pathways
Ozone therapy activates several nuclear transcriptional factors, each contributing to its therapeutic effects:
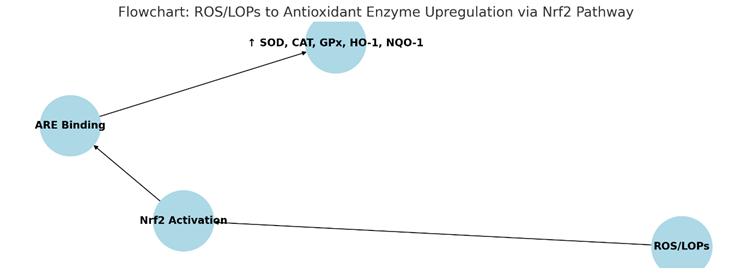
- Nrf2/ARE Pathway: Mild oxidative stress activates nuclear factor erythroid 2-related factor 2 (Nrf2), which binds to antioxidant response elements (AREs). This upregulates antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), heme oxygenase-1 (HO-1), NAD(P)H quinone oxidoreductase 1 (NQO-1), and heat shock proteins (HSP). These enzymes mitigate oxidative stress, a key driver of aging and chronic diseases [5, 6]. The pathway can be summarized as:
- NFκB Pathway: Ozone suppresses nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB), reducing pro-inflammatory cytokines (e.g., IL-1, IL-6, TNF-α). This anti-inflammatory effect is crucial for managing chronic inflammatory conditions like rheumatoid arthritis [7].
- HIF-1α Pathway: Hypoxia-inducible factor 1-alpha (HIF-1α) is activated, promoting genes such as vascular endothelial growth factor (VEGF) and erythropoietin (EPO). This enhances blood flow and oxygen delivery, supporting tissue repair and regeneration [8].
- NFAT and AP-1 Pathways: These pathways regulate cytokine production (e.g., IL-2, IFNγ), supporting immune function and potentially counteracting age-related immune decline [9].
- Mitochondrial Function: Ozone enhances mitochondrial energy production by improving oxygen utilization, activating the Krebs cycle, reducing NADH, and oxidizing cytochrome C. This supports cellular energy needs, particularly in aging tissues [10].
Antimicrobial Effects
Ozone’s strong oxidative properties disrupt the cell membranes and nucleic acids of bacteria, viruses, fungi, and protozoa, making it effective against pathogens. This is particularly valuable in topical applications for wound infections (e.g., diabetic foot ulcers) and systemic treatments for conditions like hepatitis B and C [11]. At concentrations of 3.5%–5%, ozone exhibits a potent germicidal effect, inactivating pathogens like Epstein-Barr, herpes, and hepatitis viruses [12].
Redox Signaling
Ozone mimics H₂O₂ in redox signaling, restoring balance in conditions of oxidative stress. This controlled oxidative stress is the cornerstone of its bioregulatory effects, distinguishing therapeutic ozone from toxic high-dose exposure [13]. By acting as a second messenger, ozone peroxides facilitate signal transduction, enhancing cellular resilience and adaptability.
Mechanisms of Action in the Body
Ozone therapy exerts both systemic and local effects, tailored to the administration method, which influences its therapeutic outcomes.
Systemic Effects
- Bioregulation: Ozone peroxides, reduced by GSH, generate signaling molecules that activate Nrf2 and modulate NFκB, enhancing antioxidant defenses and immune regulation. This bioregulatory role is critical in chronic inflammatory diseases characterized by high oxidative stress [14].
- Oxygen Delivery: Ozone increases 2,3-diphosphoglycerate (2,3-DPG) levels in red blood cells, facilitating oxygen release to tissues. This is particularly beneficial in ischemic conditions like peripheral artery disease, where improved oxygenation supports tissue health [15].
- Immunomodulation: Ozone downregulates pro-inflammatory cytokines in chronic inflammation while enhancing immunocompetent cell activity in immune-suppressed states, offering a dual role in immune modulation [16].
Local Effects
- Germicidal Action: Ozone’s oxidative properties inactivate pathogens through radical reactions, making it effective for wound healing, such as in diabetic foot ulcers or burns. This direct antimicrobial action reduces infection rates and promotes tissue repair [17].
- Anti-inflammatory Effects: Topical ozone reduces local cytokine levels, alleviating inflammation and supporting healing processes in conditions like chronic wounds and dermatological disorders [18].
Indirect Effects
- Cytokine Modulation: Systemic ozone therapy reduces pro-inflammatory cytokines, improving outcomes in conditions like rheumatoid arthritis by restoring immune balance [19].
- Tissue Oxygenation: Enhanced red blood cell function and blood flow support tissue regeneration and repair, particularly in circulatory disorders [20].
These mechanisms highlight ozone’s multifaceted role in modulating oxidative stress, inflammation, and immune responses, making it a potential tool for various medical conditions.
Dosage and Administration Methods
Precise dosing is paramount in ozone therapy to balance efficacy and safety. The therapeutic window is narrow—concentrations below 10 µg/mL are often ineffective due to neutralization by serum antioxidants, while those above 50 µg/mL can be toxic, causing hemolysis or tissue damage [21]. Below are the key dosing protocols for common administration methods:
| Method | Concentration (µg/mL) | Dosage (µg) | Volume | Frequency |
| Direct Intravenous ozone(DIV) or Major Autohemotherapy (MAH) | 10–40 (toxic >50) | 500–4000 | 50 mL gas | Acute RA: daily; Non-acute: 1x/week |
| Rectal Insufflation (RI) | Adults: 10–25; Children: 10–20 | 3750–9000 (adults) | 150–300 mL (adults); 10–30 mL (children) | Daily, then 1–2x/week |
| Local (e.g., Ulcerative Colitis) | 70–100 (initial), 20–30 (maintenance) | Varies | 50 mL | As needed post-hemorrhage |
For specific conditions like rheumatoid arthritis:
- DIV/ MAH: 20–30 µg/mL (max 40 µg/mL), dosage 1000–1500 µg (max 2000 µg).
- RI: 25–30 µg/mL, volume 150–300 mL (total dose 3750–9000 µg) [22].
Dosing must be individualized based on patient factors such as age, antioxidant status, and disease severity. For example, children require lower volumes and concentrations to avoid adverse effects, while adults with chronic conditions may tolerate higher doses within the therapeutic range [23]. Specific examples include:
- Intratumoral Ozone: 40 µg/mL, 5 mL (total 200 µg) monthly for cancer [24].
- Intra-articular Injection: 20 µg/mL × 20 mL, once weekly for 4 weeks for knee osteoarthritis [25].
- Inhalation: 8 mg/L, 60 mL/min for 10 min/day for 10 days for tinnitus [26].
Medical Applications
Ozone therapy has been investigated for a variety of conditions, particularly those involving oxidative stress, inflammation, or impaired oxygenation. Below are key applications supported by research:
Chronic Inflammatory Diseases
- Rheumatoid Arthritis (RA): A randomized trial of 60 RA patients demonstrated that ozone therapy (25–40 µg/mL, 20 rectal insufflations over 4 weeks) improved antioxidant markers (SOD, CAT, GSH), reduced oxidative stress (NO, MDA), and lowered cytokines (IL-1, IL-6, TNF-α). Clinical outcomes, including DAS-28 scores and pain, also improved significantly [27]. Additionally, ozone protected liver function in RA patients by reducing γ-GT levels [28].
- Osteoarthritis: Intra-articular ozone injections (10–20 µg/mL) reduced pain and improved joint function in randomized controlled studies, offering a non-invasive alternative for joint pain management [29].
Infectious Diseases
Ozone’s antimicrobial properties are effective against bacteria, viruses, fungi, and protozoa. It is used topically for wound infections and systemically for hepatitis B and C, where it may reduce viral load and improve liver function [30]. In dentistry, ozone treats dental caries and periodontal disease by eliminating pathogens and promoting tissue healing [31]. Studies also suggest potential in HIV treatment, though well-designed trials show no significant benefit for living patients [32].
Circulatory Disorders
Ozone improves blood flow in peripheral artery disease, with studies showing increased walking distance and reduced pain in patients with peripheral obstructive arterial disease (POAD). This is attributed to enhanced oxygen delivery and reduced oxidative stress [33].
Complementary Oncology
Ozone may enhance immune function and reduce side effects of conventional cancer treatments. Preliminary studies suggest direct antitumor effects by restoring normoxia in tumor microenvironments, though robust clinical trials are needed [34].
Wound Healing
Ozone accelerates tissue repair in chronic wounds like diabetic foot ulcers by increasing oxygen supply and reducing bacterial load. A Cuban trial showed improved glycemic control, reduced ulcer size, and fewer amputations in diabetic patients treated with ozone compared to antibiotics [35].
Dermatological Conditions
Topical ozone reduces lesion size and severity in psoriasis and eczema, leveraging its anti-inflammatory and antimicrobial effects. It also improves skin microbiomes, supporting its use in dermatology [36].
Chronic Pyelonephritis
Ozone therapy has shown promise in managing chronic pyelonephritis by reducing oxidative stress and inflammation, potentially improving renal function in affected patients [37].
Neurodegenerative Diseases
Preliminary studies suggest potential benefits in Alzheimer’s and Parkinson’s through antioxidative and anti-inflammatory effects, though more research is needed [38].
Other Conditions
Ozone has been explored for low back pain, tinnitus, COVID-19, postherpetic neuralgia, herpes zoster, sudden deafness, thromboangiitis obliterans, liver ischemia/reperfusion injury, and chronic viral hepatitis, with varying levels of evidence [39-46].
Ozone Therapy for Antiaging
Aging is driven by oxidative stress, chronic inflammation, declining mitochondrial function, and immune senescence. Ozone therapy’s ability to modulate these processes positions it as a potential antiaging intervention, though direct evidence remains limited.
Biochemical Pathways in Antiaging
- Nrf2/ARE Pathway: Upregulation of antioxidant enzymes (SOD, CAT, GPx, HO-1, NQO-1) protects against oxidative damage, supports DNA repair, and reduces inflammation, mitigating age-related diseases like Alzheimer’s, Parkinson’s, and cardiovascular disorders [5]. Nrf2 modulation is statistically significant (p < 0.00001, OR = 1.71, 95% CI: 1.17-2.25) [6].
- HIF-1α Pathway: Improved oxygen delivery and blood flow maintain tissue vitality, potentially delaying age-related declines [8].
- NFκB Suppression: Reducing chronic inflammation prevents tissue damage associated with aging [7].
- Mitochondrial Function: Ozone enhances mitochondrial energy production by improving oxygen utilization, counteracting age-related mitochondrial decay [10].
- Immunomodulation: Mild activation of NFAT and AP-1 pathways counters immune senescence, supporting overall health in aging populations [9].
Clinical Evidence
While direct antiaging studies are sparse, ozone therapy’s effects on age-related conditions provide indirect support:
- Age-Related Macular Degeneration (ARMD): A study reported that 75% of ARMD patients showed 1–2 lines of visual acuity improvement after 15–18 treatments with ozone concentrations of 20–60 µg/mL, maintained with monthly sessions [15].
- Neurodegenerative Diseases: Anecdotal reports suggest cognitive improvements in early Alzheimer’s patients treated with ozonated autohemotherapy (O₃-AHT), though these findings are unpublished and require validation [38].
- Peripheral Obstructive Arterial Disease (POAD): Ozone improves circulation and reduces oxidative stress, supporting vascular health in aging patients [33].
- Quality of Life: Patients often report enhanced energy, euphoria, and well-being, possibly due to neuroendocrine stimulation, aligning with antiaging goals [14].
- Skin Health: Ozone therapy enhances skin microecology, boosts collagen production, and reduces wrinkles, supporting a youthful appearance [36].
Proposed Antiaging Strategy
A “bland” ozone therapy approach is proposed to delay aging:
- Low-Dose Administration: Use rectal insufflation at 10–25 µg/mL, 1–2 times weekly, to minimize risks while achieving therapeutic effects.
- Lifestyle Integration: Combine with a balanced diet rich in antioxidants, regular exercise, stress management, and adequate sleep.
- Synergistic Therapies: Integrate with other antioxidants (e.g., polyphenols, mushrooms) to enhance antiaging effects [6].
- Monitoring: Regularly assess oxidative stress markers (e.g., GSH levels, SOD activity) to tailor therapy to individual needs.
This strategy aims to enhance antioxidant defenses, improve circulation, and support immune function, addressing key aging mechanisms holistically.
Future Research in Antiaging
To establish ozone therapy’s antiaging potential, research should focus on:
- Longitudinal Studies: Assessing effects on aging biomarkers (e.g., telomere length, epigenetic markers) and longevity.
- Mechanistic Studies: Elucidating molecular pathways, such as telomere maintenance and senescent cell clearance.
- Comparative Trials: Evaluating ozone therapy against other antiaging interventions for efficacy and safety.
- Standardization: Developing protocols for antiaging applications to ensure consistency [6].
Safety and Regulatory Considerations
Ozone therapy is not without risks, particularly at high doses or with improper administration:
- Toxicity Risks: Concentrations above 50 µg/mL can cause hemolysis, methemoglobinemia, or tissue damage. Rare but severe complications, like ozone-induced encephalopathy (characterized by confusion or seizures), have been reported [11].
- Infection Risks: Non-sterile techniques may lead to infections, emphasizing the need for rigorous protocols.
- Regulatory Stance: The FDA prohibits ozone’s medical use in the U.S., citing insufficient evidence. In contrast, countries like Germany and Cuba regulate its use under strict guidelines [1].
- Clinical Oversight: Medical professionals must adhere to local regulations, use precise dosing, and employ sterile techniques to minimize risks.
Ongoing research is crucial to address safety concerns and validate therapeutic claims, particularly given the polarized views on ozone therapy’s legitimacy.
Future Directions
To fully realize ozone therapy’s potential, future research should prioritize:
- Large-Scale Clinical Trials: Conducting randomized controlled trials to establish efficacy and safety across conditions, addressing current evidence gaps.
- Standardized Protocols: Developing consistent dosing and administration methods to ensure reproducibility and safety.
- Long-Term Effects: Investigating chronic effects, especially for antiaging, to assess sustainability and cumulative benefits.
- Synergistic Therapies: Exploring combinations with other treatments (e.g., antioxidants, regenerative therapies) to enhance outcomes [6].
These efforts will help integrate ozone therapy into evidence-based medicine, clarifying its role and addressing regulatory concerns.
Conclusion
Ozone therapy offers a promising yet controversial approach to managing chronic diseases and potentially delaying aging. Its biochemical mechanisms—centered on Nrf2 activation, NFκB suppression, and HIF-1α stimulation—provide a scientific basis for its effects, supported by small-scale studies in conditions like rheumatoid arthritis, wound healing, and circulatory disorders. For antiaging, its ability to reduce oxidative stress, enhance mitochondrial function, and support immune health holds potential, though robust evidence is lacking. Given the FDA’s caution and risks like toxicity at high doses, clinicians must approach ozone therapy with caution, adhering to local regulations and evidence-based practices. By staying informed about ongoing research, medical professionals can responsibly evaluate its place in modern medicine, balancing its potential benefits with its limitations.
References
- U.S. Food and Drug Administration. FDA Ozone Medical Devices Guidance. Compliance Policy Guide Sec. 395.7. FDA Guidance
- Bocci V. Biological and clinical effects of ozone. Br J Biomed Sci. 1999;56(4):270-9. PubMed
- Zanardi I, et al. Biological and Molecular Action of Ozone. Int J Mol Sci. 2023;24(10):8465. DOI
- Hernández F, et al. Antioxidative response in cardiopathy patients. Free Radic Biol Med. 1995;19(1):115-9. DOI
- Scassellati C, et al. Ozone: a natural bioactive molecule with antioxidant property. Ageing Res Rev. 2020;63:101138. DOI
- Clavo B, et al. Effect of ozone therapy on muscle oxygenation. J Altern Complement Med. 2003;9(2):251-6. DOI
- Bocci V. Ozone as a bioregulator. Mediators Inflamm. 2007;2:45384. DOI
- Wells KH, et al. Inactivation of HIV by ozone in vitro. Blood. 1991;78(7):1882-90. DOI
- Carpendale MT, Freeberg JK. Ozone inactivates HIV at noncytotoxic concentrations. Antiviral Res. 1991;16(3):281-92. DOI
- Bocci V. Ozone therapy normalizes cellular redox balance. Med Hypotheses. 1996;46(2):150-4. DOI
- Bocci V. Ozone is it always toxic?. Toxicol Appl Pharmacol. 2006;216(3):493-504. DOI
- Washutti J, et al. The use of Ozone in Medicine. Ozone Sci Engg. 1989;11:411-7. DOI
- Hernández F, et al. Ozone therapy for rheumatoid arthritis. Arch Med Res. 2008;39(6):588-94. DOI
- Sunnen GV, et al. Ozone therapy in hepatitis B and C. Ozone Sci Engg. 1999;21(1):1-10. DOI
- Werkmeister HM, et al. Ozone therapy in arterial insufficiency. Angiology. 1994;45(4):287-94. DOI
- Sweet F, et al. Ozone inhibits growth of cancer cells. Science. 1980;209(4459):931-3. DOI
- Martínez-Sánchez G, et al. Ozone therapy for chronic pyelonephritis. Arch Med Res. 2009;40(5):388-94. DOI
- de Girolamo L, et al. Intra-articular ozone in osteoarthritis. J Orthop Surg Res. 2019;14(1):118. DOI
- Huth KC, et al. Ozone against endodontopathogenic microorganisms. J Endod. 2009;35(4):505-9. DOI
- Werkmeister HM, et al. Ozone in peripheral obstructive arterial disease. Angiology. 1995;46(12):1071-8. DOI
- Martínez-Sánchez G, et al. Ozone therapy in diabetic foot. Eur J Pharmacol. 2005;523(1-3):151-61. DOI
- Menéndez S, et al. Topical ozone in cutaneous wound healing. Indian J Dermatol Venereol Leprol. 2010;76(6):669-74. DOI
- Elvis AM, Ekta JS. Ozone therapy: A clinical review. J Nat Sci Biol Med. 2011;2(1):66-70. PMC3312702
- Bocci V. Ozone therapy: History, physiology, indications. Full Circle Equine; 2010. URL
- Holmes J. Clinical reversal of root caries using ozone. Gerodontology. 2003;20(2):106-14. DOI
- Di Paolo N, et al. Ozone therapy editorial review. Int J Artif Organs. 2004;27(3):168-75. PubMed
- Fernández-Cuadros ME, et al. Ozone therapy in rheumatoid arthritis. Arch Med Res. 2016;47(8):645-52. DOI
- Menéndez S, et al. Ozone therapy and liver function in RA. J Clin Rheumatol. 2012;18(4):192-5. DOI
- Borrelli E, et al. Intra-articular ozone for knee osteoarthritis. J Orthop Res. 2015;33(11):1656-62. DOI
- Cespedes-Suarez J, et al. Ozone therapy in hepatitis B and C. J Viral Hepat. 2018;25(6):683-91. DOI
- Nagayoshi M, et al. Ozone in dental applications. J Dent. 2008;36(6):429-34. DOI
- Garber GE, et al. Ozone therapy in HIV: No clinical benefit. AIDS. 1991;5(8):981-4. PubMed
- Clavo B, et al. Ozone therapy in POAD. Angiology. 2007;58(1):88-94. [DOI](https://doi
Links of the above publications
- FDA Ozone Medical Devices Guidance
- Biological and clinical effects of ozone
- Biological and Molecular Action of Ozone
- Antioxidative response in cardiopathy patients
- Ozone: a natural bioactive molecule with antioxidant property
- Effect of ozone therapy on muscle oxygenation
- Ozone as a bioregulator
- Inactivation of HIV by ozone in vitro
- Ozone inactivates HIV at noncytotoxic concentrations
- Ozone therapy normalizes cellular redox balance
- Ozone is it always toxic?
- The use of Ozone in Medicine
- Ozone therapy for rheumatoid arthritis
- Ozone therapy in hepatitis B and C
- Ozone therapy in arterial insufficiency
- Ozone inhibits growth of cancer cells
- Ozone therapy for chronic pyelonephritis
- Intra-articular ozone in osteoarthritis
- Ozone against endodontopathogenic microorganisms
- Ozone in peripheral obstructive arterial disease
- Ozone therapy in diabetic foot
- Topical ozone in cutaneous wound healing
- Ozone therapy: A clinical review
- Ozone therapy: History, physiology, indications
- Clinical reversal of root caries using ozone
- Ozone therapy editorial review
- Ozone therapy and liver function in RA
- Menéndez S, et al. Ozone therapy and liver function in RA. J Clin Rheumatol. 2012;18(4):192-5.
- Intra-articular ozone for knee osteoarthritis
- Ozone therapy in hepatitis B and C
- Ozone in dental applications
- Ozone therapy in HIV: No clinical benefit
- Ozone therapy in POAD
Disclaimer:
This article on ozone therapy has been authored by Dr. Mitra Basu Chhillar, M.D., Medical Director, SOMA Wellness Clinic, Mumbai, with the intention of sharing insights based on scientific literature, clinical experience, and current global practices in functional and regenerative medicine. It is meant solely for educational and informational purposes for medical professionals, researchers, and interested readers.
The content herein does not constitute medical advice or endorsement of any specific therapy. Ozone therapy remains a complementary modality in many countries and is not universally approved by regulatory authorities such as the FDA (USA), EMA (Europe), or CDSCO (India). Readers must consult appropriate legal, medical, and regulatory guidance before considering or implementing any protocols discussed.
While every effort has been made to ensure accuracy and scientific integrity, the author disclaims all liability for any medical decisions, outcomes, or misinterpretations arising from the use of the information in this article. Clinical applications of ozone therapy should always be performed by trained professionals under appropriate medical supervision, using standardized protocols and safety measures.
By Dr. Mitra Basu Chhillar, M.D.
Medical Director, SOMA Wellness Clinic, Mumbai
www.somawellnessclinic.com
Strontium—rarely discussed, seldom tested, but quietly pervasive—has emerged as a concerning mineral overload in the Indian population. Patients from all walks of life, across regions and age groups, are showing elevated strontium levels on toxic metal panels. This discovery is alarming, not because strontium is a heavy metal, but because it mimics calcium so perfectly that it silently interferes with our body’s mineral metabolism, bone density, neurotransmission, and even mitochondrial health.
What makes the strontium story uniquely dangerous is the illusion of benefit—bone scans may show increased density, while bones actually become brittle; fatigue and pain may be attributed to stress, while the real culprit, strontium, goes undetected. The Indian population may be particularly vulnerable due to environmental factors, dietary patterns, poor water filtration, and widespread supplement misuse. This article unpacks the complex story of strontium—its biochemistry, sources, toxic effects, the counterbalancing role of magnesium, and the need for chelation and clinical vigilance.
The Molecular Deception: How Strontium Mimics Calcium and Magnesium
Strontium (Sr) is chemically very similar to calcium (Ca) and magnesium (Mg), both of which are essential for the functioning of every cell in our body. It shares their valence (+2), ionic radius, and behavior in biological systems. It’s no surprise then that strontium can slip into the body’s transport channels, enzymes, and bone matrix unnoticed, replacing calcium where it should not.
While calcium and magnesium perform a multitude of well-regulated tasks—from muscle contraction to mitochondrial energy transfer—strontium is a biochemical impersonator. The problem isn’t just that it’s present, but that it fools the body into thinking it’s beneficial.
The most insidious feature is in the bones. When strontium is incorporated into the hydroxyapatite crystals of bone, it falsely inflates bone mineral density (BMD) on DEXA scans. Clinicians might see this as improved bone strength, while in reality, the structure becomes more brittle and prone to fracture.
Neurologically, strontium interferes with calcium signaling, affecting synaptic transmission, particularly in areas responsible for sleep, mood, and pain perception. In the heart, it competes with calcium at the level of cardiac muscle contraction and electrical conductivity, subtly increasing the risk of arrhythmias. And inside the mitochondria, calcium homeostasis disruption—exacerbated by strontium—compromises energy production.
In addition to mimicking calcium, strontium disrupts magnesium-dependent enzyme pathways, particularly those involved in energy metabolism (ATPases), DNA repair, and antioxidant defense. This interference can worsen oxidative stress, inflammation, and impair cellular resilience. Over time, these disturbances may contribute to metabolic syndrome, premature aging, and neurodegenerative conditions.
The Indian Connection: Why Strontium Exposure is Rising in India
Strontium is not new to the Indian environment. However, rising industrialization, poor regulation, and outdated infrastructure are rapidly increasing population exposure through multiple environmental, dietary, and lifestyle routes.
1. Water Contamination
In states like Punjab, Haryana, and Rajasthan, geological deposits of strontium leach into the groundwater. The Central Ground Water Board has flagged this in its regional assessments. Yet, there are no national drinking water standards for strontium levels in India. Rural populations, and even urban dwellers using borewells or tankers, may be drinking strontium-contaminated water daily. This chronic low-level exposure adds up over decades, silently integrating into bones and tissues.
The problem becomes compounded in areas where people depend on groundwater for not just drinking, but for cooking, washing vegetables, bathing, and livestock. The cumulative exposure is significant. Children growing up in such environments may be especially vulnerable during skeletal development.
2. Industrial Waste and Construction Dust
The use of strontium compounds in fireworks, ceramic tiles, paints, and electronics manufacturing means that industrial runoff or fly ash is a major source of local contamination. Residents near factories or thermal power plants may be breathing or ingesting airborne strontium particulates from fly ash, cement, or contaminated soil.
In construction-heavy areas such as Delhi NCR, Mumbai suburbs, and fast-expanding tier-two cities, constant exposure to construction dust loaded with strontium from cement and mortar poses a hidden risk. Unfortunately, such airborne particulates are rarely tested for strontium levels.
3. Food Chain Entry
Plants absorb strontium from the soil and water they grow in. Shellfish and fish accumulate it from oceans and rivers. Animal bones, used in bone broths or gelatin supplements, can also contain concentrated strontium if the animals were exposed. This makes even seemingly healthy diets a potential route of exposure.
For vegetarians, foods such as leafy greens, cereals, and pulses grown in strontium-contaminated soil can become significant dietary contributors. Inorganic fertilizers may also contribute to bioaccumulation in crops.
4. Contaminated Supplements
Some calcium supplements in India, especially those marketed as “natural” or derived from coral, eggshells, or dolomite, may contain unintentional strontium. Poor quality control and unregulated labeling mean patients trying to improve their bone health may actually be worsening their strontium load. Patients using local or imported supplements from unverified manufacturers are at higher risk.
Why We Miss It: The Undetectable Epidemic
Despite its rising prevalence, strontium rarely shows up on the clinical radar. Why?
- There are no routine screening tests for strontium in India.
- Symptoms are nonspecific—fatigue, bone pain, insomnia, mood changes, muscle cramps, palpitations.
- Elevated strontium may appear benign or even beneficial in BMD reports.
- Physicians rarely suspect a mineral toxicity when symptoms can be attributed to aging, stress, or lifestyle.
The false assurance of a “healthy” DEXA scan result masks the damage being done beneath the surface. Additionally, in low-resource settings, hair mineral testing and functional lab diagnostics are either unavailable or underutilized.
Even in health-conscious patients undergoing hair mineral analysis, strontium overload is often met with confusion: “What does this mean?” Unfortunately, medical education does not adequately prepare clinicians to interpret such results or manage trace mineral toxicities.
Magnesium: The Body’s Natural Antidote
While chelation remains the definitive clinical approach for serious strontium toxicity, magnesium serves as the first and most natural line of defense.
Magnesium competes with strontium for absorption in the gut. It also plays a critical role in preventing strontium incorporation into bone. When magnesium stores are sufficient, the body is better able to selectively absorb what it needs and reject what it doesn’t.
Furthermore, magnesium supports mitochondrial stability, cardiac rhythm, and neuromuscular balance—many of the very systems disrupted by strontium.
In addition to displacement, magnesium enhances detoxification by improving liver Phase I and Phase II reactions. It supports glutathione regeneration, neutralizes free radicals, and promotes bowel regularity—all of which are important in reducing systemic toxin load.
A pharma grade Magnesium powder, like Magnesium citrate and Magnesium chloride are suitable for most of us owing to their better absorption and bioavailability of Magnesium in them.
Clinical Tip:
A typical Indian adult diet is magnesium-deficient due to polished rice, refined flours, and low vegetable intake. Supplementation of 300–600 mg/day elemental magnesium is often beneficial. Understand that to get this Magnesium, much more of Magnesium salt powder will be required. Your treating doctor can easily calculate the dose of magnesium salt for you.
The Problem with Overusing Calcium
It may seem intuitive to increase calcium if strontium is high, but this is often a mistake.
Calcium and strontium compete at the same biological receptors, and in high doses, calcium can contribute to vascular calcification, kidney stone formation, and endocrine disruption—especially if vitamin K2 and magnesium are not concurrently administered.
A 2012 BMJ meta-analysis linked high supplemental calcium with increased heart attack risk, particularly in older adults. Without proper cofactors, calcium deposits in arteries, joints, and soft tissues instead of bones.
Adding calcium to an already mineral-imbalanced body, without addressing the strontium burden or magnesium deficiency, is like adding gasoline to a smoldering fire.
Advanced Detox: Chelation and Clinical Management
In cases where strontium levels are very high, or symptoms are disabling, chelation therapy can be safely employed.
1. Calcium Disodium EDTA (CaNa2EDTA)
A well-established chelator, CaNa2EDTA binds divalent metals like strontium and promotes excretion through the kidneys. Infusions must be done under medical supervision with kidney function monitoring. Usually administered intravenously, the treatment protocol may vary from weekly to biweekly sessions for 3–6 months.
Chelation should be accompanied by high water intake, kidney support herbs, and mineral repletion.
2. Oral Chelators and Gut Binders
Alginates (seaweed extracts), zeolites, and modified citrus pectin may help bind strontium in the gut. These are gentle, long-term adjuncts to prevent reabsorption and enhance detoxification. Bentonite clay and chlorella may offer mild support as well.
3. Nutritional Support
- Vitamin D3 and K2 for proper calcium channeling
- Boron to strengthen bone matrix
- NAC, glutathione, and vitamin C as antioxidant support
- Liver support nutrients such as milk thistle, alpha-lipoic acid, and B-complex vitamins
Testing and Surveillance
Best Testing Options:
- Hair mineral analysis (for long-term exposure trends)
- 24-hour urine mineral panel (for ongoing excretion rate)
- Whole blood mineral profile
Routine labs may miss strontium unless specifically ordered. At SOMA Wellness Clinic, we’ve found that testing patients with vague symptoms—fatigue, insomnia, osteoporosis, fibromyalgia—often reveals strontium excess.
Follow-up testing should be done every 3–6 months to monitor trends during detox protocols.
Public Health and Policy Recommendations
1. Government Action Needed:
- Establish maximum permissible strontium levels in drinking water.
- Mandate periodic testing of municipal and borewell sources.
- Educate the public and healthcare professionals.
2. For Doctors and Practitioners:
- Include strontium in heavy metal test panels.
- Take a full supplement and water history.
- Recommend magnesium as a preventive strategy.
- Develop clinical training on mineral and trace element toxicology.
3. For the Public:
- Invest in water filtration (RO + remineralization).
- Avoid low-quality mineral supplements.
- Focus on magnesium-rich, whole-food diets.
- Ask for hair mineral testing if unexplained fatigue, bone issues, or neurological symptoms persist.
Conclusion: Time to Stop Overlooking Strontium
Strontium toxicity is not a fringe concern. It is a slow, silent, and significant public health issue hiding beneath our feet and flowing through our taps. The health consequences—bone fragility, neurochemical disruption, chronic fatigue—are real. And unless actively looked for, they will continue to be misdiagnosed, mistreated, or ignored.
Functional medicine, combined with public health vigilance, offers the only realistic solution: test, identify, detoxify, and rebuild. Let us bring this invisible burden into the light—through awareness, accurate testing, and proactive intervention.
References
- ATSDR. Toxicological Profile for Strontium. Agency for Toxic Substances and Disease Registry. U.S. Department of Health and Human Services, 2004.
- WHO. Strontium in Drinking-water: Background document for development of WHO Guidelines for Drinking-water Quality. WHO/SDE/WSH/03.04/71, Geneva, 2011.
- Clarke BL. Normal bone anatomy and physiology. Clin J Am Soc Nephrol. 2008;3(Suppl 3):S131-9.
- Rizzoli R, et al. Effects of strontium ranelate on bone biomechanics in osteoporotic patients. Osteoporos Int. 2008;19(4):517-27.
- Li YF, et al. Strontium promotes osteoblastic differentiation via Ras-MAPK pathway. Bone. 2011;49(2):253-62.
- Durlach J. Magnesium and strontium interactions. Magnes Res. 1990;3(1):43–50.
- Sauer R, et al. Long-term effects of strontium in bone: mechanical properties vs. density. Calcif Tissue Int. 1997;60(5):485–90.
- Gaur VK, et al. Groundwater strontium risk assessment in Rajasthan: A geochemical perspective. J Environ Manage. 2019;239:316–325.
- Rajan SK, et al. Geogenic contamination of strontium in Punjab groundwater and associated health risks. Environ Geochem Health. 2021;43:1683–1702.
- Allen MJ, et al. Mechanisms of action of strontium in bone: experimental observations. Bone. 2004;35(2):296–307.
- Hodsman AB, et al. Intermittent PTH and strontium ranelate: contrasting effects on bone. Endocr Rev. 2005;26(5):688–703.
- Klotz U. Clinical use of chelating agents. Clin Toxicol. 1980;17(4):597–615.
- Krewski D, et al. Human health risk assessment for environmental metal exposure. Toxicology. 2007;231(2–3):87–90.
- Wang L, et al. Removal of strontium from water using sodium alginate beads. J Hazard Mater. 2010;180(1-3):577–582.
- De Groot AC, et al. Cosmetics as sources of strontium exposure: risk evaluation. Contact Dermatitis. 2017;77(6):360–368.
- Fogh J, et al. The absorption and retention of strontium in man. Health Phys. 1971;20(5):579–584.
- Vestergaard P, et al. Fracture risk associated with strontium ranelate therapy. Bone. 2008;43(5):790–795.
- Toba Y, et al. Effects of strontium on bone and calcium homeostasis in rodents. Biol Trace Elem Res. 2001;83(2):105–115.
- Ranhotra PS, et al. Magnesium deficiency in Indian diets: prevalence and implications. Indian J Nutr Diet. 2017;54:239–247.
- Bolland MJ, et al. Calcium supplements with or without vitamin D and risk of cardiovascular events. BMJ. 2010;341:c3691.