Depression is frequently discussed in low tones. Many people carry it discreetly while still meeting the demands of their jobs, families, and daily lives. Despite this quiet, one of our time’s most serious public health concerns remains. According to the World Health Organization, depression affects hundreds of millions of people globally and is still a primary cause of disability.
For many people, treatment with psychotherapy and antidepressants delivers significant alleviation. Despite obtaining appropriate care, a large percentage of people continue to struggle. In recent years, physicians and researchers have begun investigating new therapies that may benefit patients who have not received relief from traditional approaches. Among these developing alternatives, ketamine therapy has received a lot of interest in psychiatric care.
Understanding What is Depression
To grasp the significance of developing medicines, ask a simple question: What is depression?
Depression is a medical disorder that affects one’s mood, thought patterns, energy levels, and physical health. It is more than just a period of grief or a bad week. Clinical depression is characterised by long-term changes in a person’s feelings, thoughts, and functioning.
Depression is sometimes referred to in medical terms as major depressive disorder, a condition marked by a persistent poor mood or a loss of interest in previously significant activities. These alterations can have an impact on sleep, hunger, attention, and enjoyment.
Psychiatric researchers consider depression to be a complicated illness involving several biological and psychological systems. Brain chemistry, genetics, stress exposure, and life events all have an impact. Neurotransmitters, including serotonin, dopamine, and glutamate, affect how brain cells communicate. When these systems are out of balance, emotional control and motivation might suffer.
Depression can strike at any stage of life. Some people have only one episode, while others have repeated cycles that last for years. Early diagnosis of the disease might assist patients in seeking appropriate assistance and treatment.
Recognising the Symptoms of Depression
The symptoms of depression often develop gradually. At first, they may appear as subtle shifts in mood or energy. Over time, they can begin to affect daily functioning.
Common depression symptoms include:
- Persistent sadness or low mood
- Loss of interest in activities that were once enjoyable
- Fatigue or reduced energy
- Changes in sleep patterns, including insomnia or excessive sleeping
- Changes in appetite or body weight
- Difficulty concentrating or making decisions
- Feelings of guilt, hopelessness, or worthlessness
- Slower movement or agitation
- Social withdrawal
- Thoughts of self-harm or suicide in severe cases
These signs of depression vary from person to person. Some individuals experience primarily emotional symptoms, while others notice physical changes such as exhaustion, headaches, or digestive discomfort.
It is also possible for depression to exist alongside anxiety, trauma-related conditions, or chronic stress. Many people continue to function professionally while experiencing internal emotional numbness or detachment. This presentation is sometimes described informally as high-functioning depression.
Recognising these patterns early can allow individuals to access appropriate care before symptoms deepen.

Conventional Treatments for Depression
Modern psychiatry offers several evidence-based treatments for depression. These typically include:
Psychotherapy
Therapies such as cognitive behavioural therapy and trauma-informed counselling help individuals understand thought patterns and develop healthier coping strategies.
Antidepressant medication
Medications such as selective serotonin reuptake inhibitors aim to stabilise neurotransmitter systems that influence mood regulation.
Lifestyle and behavioural interventions
Exercise, sleep regulation, social connection, and stress management all play supportive roles in recovery.
For many individuals, these treatments offer meaningful improvement. However, approximately one-third of people with depression experience what clinicians describe as treatment-resistant depression, where symptoms persist despite multiple treatment attempts.
This challenge has prompted researchers to explore new pharmacological approaches that work through different biological pathways.
The Emergence of Ketamine Therapy
Ketamine has been used in medicine for decades as an anaesthetic. It was first developed in the 1960s and later approved for surgical use because of its reliable safety profile in controlled clinical environments.
In the early 2000s, psychiatric researchers began observing something unexpected. Patients receiving low doses of ketamine during clinical studies reported rapid improvements in depressive symptoms. These findings sparked a wave of research into ketamine therapy for depression.
Today, ketamine is being studied and used in specialised clinical settings for individuals who have not responded fully to conventional treatments. It is important to emphasise that therapeutic ketamine is administered under strict medical supervision with carefully calibrated protocols.
The goal is not sedation but the creation of a temporary neurological state that allows the brain to reorganise emotional patterns.
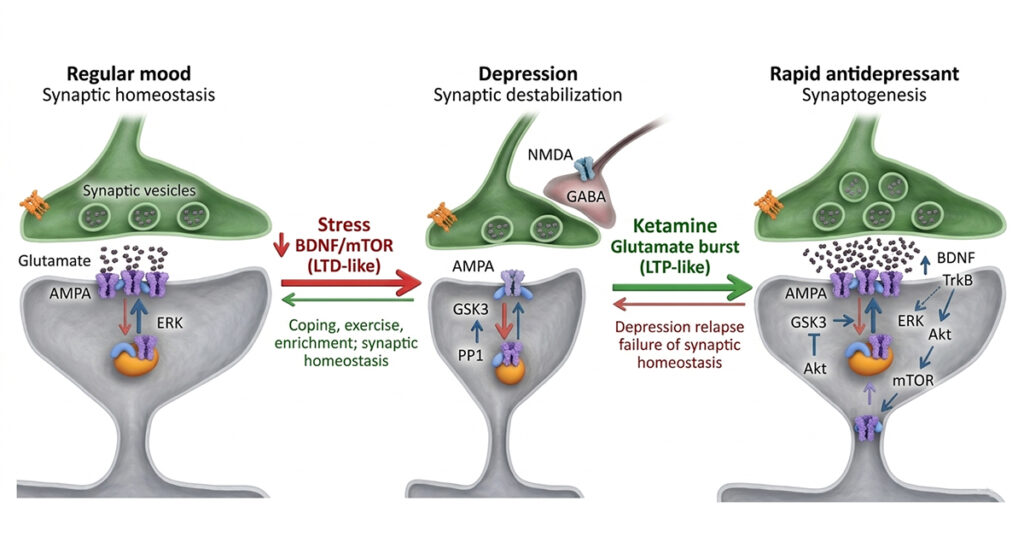
How Ketamine Affects the Brain
Traditional antidepressants typically influence the serotonin and norepinephrine systems. Ketamine works via a distinct method that involves the brain’s glutamate signalling network.
Glutamate is the most common excitatory neurotransmitter in the human brain. It is crucial for learning, memory, and brain transmission.
Research shows that ketamine temporarily blocks NMDA receptors in this system. This action initiates a series of biological events that enhance glutamate release and promote the development of new synaptic connections.
Simply put, ketamine may aid the brain in the formation of new neural communication pathways. Scientists refer to this ability as neuroplasticity.
According to studies by institutes such as the National Institute of Mental Health, increased neuroplasticity may help individuals absorb emotions and experiences in novel ways, particularly when therapy is incorporated into treatment.
These biochemical changes may explain why some people experience changes in mood, perspective and emotional flexibility after receiving carefully monitored treatment.
Understanding Ketamine Effects in Clinical Care
The effects of ketamine during therapeutic sessions differ from person to person. In a regulated clinical setting, the medicine is usually given in modest dosages by intravenous infusion, oral formulations, or other medically supervised delivery techniques.
These doses are substantially lower than those used for anaesthesia.
During treatment, some people report experiences like these:
- A sense of alienation from normal mental patterns
- Enhanced introspection.
- Visual or sensory alterations
- Emotional insights
These effects are transient and typically disappear within a few hours. Medical staff monitors patients throughout the session to ensure their safety and comfort.
Researchers believe that psychological experience, paired with biological changes in neuroplasticity, may open up a window for more effective therapeutic reflection.
Ketamine Dose and Treatment Protocols
Determining the appropriate ketamine dose requires careful medical evaluation. Factors such as medical history, current medications, and mental health profile are all considered before treatment begins.
Clinical protocols generally involve a series of low-dose sessions administered over several weeks. Each session is supervised by trained medical professionals who monitor vital signs and patient well-being.
Because ketamine influences brain chemistry and perception, responsible clinics follow strict screening procedures. This ensures that the treatment is suitable for the individual and that risks are minimised.
The field of ketamine assisted therapy continues to evolve as new clinical trials expand our understanding of optimal dosing strategies and long-term outcomes.
Ketamine Therapy in a Therapeutic Setting
While the pharmacological effects of ketamine are important, many clinicians emphasise the role of the surrounding therapeutic environment.
The treatment setting often includes psychological support, integration sessions, or reflective practices that help individuals process insights that may arise during treatment.
This integrative approach acknowledges that depression involves emotional patterns, life experiences, and neurological processes. Addressing all of these elements can help create a more supportive pathway toward recovery.
Our Approach at Soma Wellness Clinic
At Soma Wellness Clinic, we approach ketamine therapy with careful clinical oversight and a deep respect for the complexity of mental health.
Our protocol uses low-dose ketamine therapy in a structured medical setting designed to support emotional reset. Each treatment is conducted under the supervision of qualified physicians and follows applicable medical and regulatory standards.
We see ketamine as a therapeutic tool rather than a standalone solution. When appropriate, it is paired with psychotherapy, journaling, breathwork, or reflective practices. This integrated approach helps individuals explore the insights that may arise during treatment and translate them into lasting emotional change.
Our programme is designed for individuals who may be facing:
- Anxiety or trauma-related challenges
- High-functioning depression or emotional numbness
- Limited response to traditional talk therapy or antidepressant medication
Within a guided timeframe, carefully measured microdoses are administered in a safe clinical environment. The intention is to support neuroplasticity and allow the brain to develop healthier adaptive pathways.
Many individuals describe a renewed ability to access feelings of connection, clarity, and emotional flexibility after treatment. Progress can sometimes emerge within weeks, although each person’s experience remains unique.
Our role is to provide a supportive and medically responsible environment in which individuals can explore this emerging therapeutic pathway.
A Developing Field in Mental Health Care
Ketamine therapy has introduced a promising direction in the treatment of depression. By acting on different neural pathways and supporting neuroplasticity, it has expanded how clinicians understand and approach difficult to treat depression.
At the same time, this therapy requires careful screening, precise dosing, and medical supervision. Responsible clinical settings combine ketamine treatment with psychological support to ensure safety and meaningful outcomes. For individuals who have struggled with persistent depression, these advances represent a new possibility in mental health care.
At Soma Wellness Clinic, we offer medically supervised ketamine therapy designed to support emotional healing in a safe and structured environment. If you would like to explore whether this approach may be suitable for you, our team is here to help.
References
- World Health Organization
https://www.who.int/news-room/fact-sheets/detail/depression
- National Institute of Mental Health
https://www.nimh.nih.gov/health/topics/depression
- Cleveland Clinic Ketamine Therapy
https://my.clevelandclinic.org/health/treatments/ketamine-therapy
- National Institutes of Health – Mechanisms of Ketamine in Depression
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7225830
- Depression Overview
https://en.wikipedia.org/wiki/Major_depressive_disorder
- Mental Health Conditions: Depression and Anxiety
https://www.cdc.gov/tobacco/campaign/tips/diseases/depression-anxiety.html
- Ketamine Medical Overview
https://en.wikipedia.org/wiki/Ketamine
- Harvard Health Publishing: Ketamine for treatment-resistant depression: When and where is it safe?
- University of Utha: FAST FACTS ABOUT KETAMINE FOR DEPRESSION
https://healthcare.utah.edu/healthfeed/2026/01/fast-facts-about-ketamine-depression
- Yale Medicine: Ketamine for Depression: How It Works
https://www.yalemedicine.org/news/ketamine-for-depression
- Mediclinic: Ketamine infusions: a breakthrough treatment for some patients with depression
FAQs
1. How does ketamine therapy help with depression?
Ketamine works differently than other antidepressants. It affects the brain’s glutamate system and may aid in the formation of new synaptic connections. This method can help some people improve their emotional flexibility and minimise their depression symptoms.
2. Who can consider ketamine therapy for depression?
Ketamine therapy is often explored by people who have not seen enough improvement with antidepressants or talk therapy. A doctor will always assess whether this treatment is suitable before starting.
3. What happens during a ketamine therapy session?
In a clinical setting, a small and carefully monitored ketamine dose is given under medical supervision. Some people feel relaxed or reflective during the session. The ketamine effects usually wear off within a few hours.
4. Is ketamine therapy safe?
When done in a medical clinic with trained professionals, ketamine therapy for depression is carefully monitored and controlled. Doctors screen patients, manage the dose, and observe the entire session to ensure safety.
Time moves quietly. It leaves no visible trail as it passes. Yet the body keeps an account.
Lines appear. Energy shifts. Sleep feels different. Recovery takes longer than it once did. These changes form a pattern that most people simply call ageing.
For many years, age was treated as a number tied to a date of birth. Scientific research now separates chronological age from the biological age. Chronological age counts years. Biological age reflects how well the body is functioning at a cellular level.
In 2013, researchers described what they called the “hallmarks of ageing” in the journal Cell. These hallmarks include DNA damage, mitochondrial decline, cellular senescence, and chronic inflammation. These processes develop gradually, yet their speed varies between individuals.
Two people of the same age can show very different biological profiles. One may have resilient metabolic markers and strong recovery capacity. The other may display early strain in similar systems. This variation has led scientists to examine the idea of premature ageing.
What Is Premature Ageing?
Premature ageing refers to biological changes that occur earlier or progress more rapidly than expected for a given chronological age. It concerns internal processes rather than appearance alone.
One widely studied indicator of ageing is telomere length. Telomeres sit at the ends of chromosomes and protect genetic material during cell division. With each division, telomeres shorten slightly. Over time, this shortening contributes to cellular ageing. Research published in PNAS demonstrated that chronic psychological stress was associated with shorter telomeres. This finding suggested that lifestyle and environment can influence biological ageing.
Another development in ageing science is the epigenetic clock. In 2013, Steve Horvath introduced a method of estimating biological age by analysing patterns of DNA methylation. These chemical modifications act like markers that reflect cellular history. When methylation age exceeds chronological age, it may indicate accelerated ageing.
Inflammation also plays a central role. Scientists use the term inflammaging to describe the gradual rise in inflammatory activity that accompanies ageing. Persistent low-grade inflammation contributes to cardiovascular disease, metabolic dysfunction, and cognitive decline.
Premature ageing therefore reflects a shift in how quickly these biological mechanisms unfold.
Signs That Ageing May Be Accelerating
Ageing rarely arrives in a dramatic way. It tends to settle in quietly. A little less energy in the morning. Muscles that feel slower to recover. An afternoon slump that did not exist a few years ago. The changes are small, yet they linger.
Ongoing fatigue is one of the most common concerns after the age of thirty. It is different from feeling tired after a late night. This kind of fatigue can sit in the background even after rest. The World Health Organization recognises chronic fatigue syndrome as a medical condition, yet many people experience steady exhaustion without meeting formal diagnostic definitions. Scientific studies have explored possible explanations, including low-grade inflammation and reduced efficiency in the body’s energy systems.
Energy production happens inside cells, within structures called mitochondria. They act as microscopic power stations. As mitochondrial performance declines with age, the supply of cellular energy can decrease. Research published in Molecular Cell describes mitochondrial dysfunction as a central feature of the ageing process. When energy becomes less available, ordinary tasks can begin to feel unexpectedly demanding.
Hormonal shifts add another dimension. During perimenopause, levels of oestrogen and progesterone gradually fluctuate. In men, age-related testosterone decline, often referred to as andropause or male menopause, may influence strength, mood, and vitality. A study in the New England Journal of Medicine reported associations between lower testosterone levels and reduced physical function in older men.
Sleep often changes alongside these hormonal adjustments. Falling asleep may take longer. Waking during the night becomes more common. Research has linked shorter sleep duration with reduced telomere length and increased inflammatory markers. Over time, broken sleep can place additional pressure on already strained biological systems.
Each of these signs can occur for many reasons. When several appear together and persist, they may suggest that the body is carrying a heavier biological load than expected for its age.
Why Does Ageing Accelerate?
The causes of ageing involve overlapping pathways rather than a single trigger.
Chronic stress influences hormonal balance and immune function. Elevated cortisol over extended periods can affect metabolic stability and tissue repair. The earlier study on stress and telomeres highlighted this connection.
Oxidative stress provides another explanation. During normal metabolism, cells produce reactive oxygen species. In balanced amounts, these molecules are manageable. When antioxidant defences fall behind, oxidative damage accumulates. A review in Clinical Interventions in Ageing outlined the link between oxidative stress and age-related disease.
Lifestyle factors matter. Sedentary behaviour reduces mitochondrial density and metabolic efficiency. Exercise research consistently shows improvements in mitochondrial function following regular physical activity.
Hormonal shifts further shape ageing trajectories. Oestrogen and testosterone influence bone density, muscle mass, mood regulation, and fat distribution. Gradual decline can alter physical and cognitive resilience.
Ageing, therefore, reflects cumulative biological wear. When stress, metabolic imbalance, and hormonal shifts converge, early ageing may emerge.
Senescent Cells and the Biology of Ageing
As cells are damaged, they enter a state known as senescence. Senescent cells stop dividing yet remain physiologically active. They release inflammatory chemicals into the surrounding tissues.
According to studies published in Nature Medicine, the accumulation of senescent cells causes tissue dysfunction and persistent inflammation. Removing these cells from animal models improved physical health markers. Human trials are still ongoing, and long-term results are being examined.
Senescent cells have become a hot topic in longevity research. Scientists are still looking at whether targeting these cells can promote healthier ageing patterns under good medical care.
Can Biological Age Be Influenced?
Ageing is a natural biological process, yet the rate at which it occurs varies greatly across individuals. According to longevity science research, biological ageing is regulated by various interconnected systems in the body, such as metabolism, hormones, inflammation and cellular energy generation.
Physical activity plays a major role in maintaining these systems. Exercise helps improve mitochondrial function, supports insulin sensitivity, and preserves muscle mass, which naturally declines with age.
Nutrition also contributes to how the body adapts over time. Diets centred on whole foods, fibre, and healthy fats are associated with healthier metabolic and inflammatory markers. Sleep is equally important. Consistent and restorative sleep allows the body to regulate hormones and repair cellular damage that accumulates during daily life.
Hormonal changes can also shape how ageing is experienced. Shifts in oestrogen, progesterone, or testosterone may influence energy levels, recovery, mood, and metabolism. When these changes become noticeable, a structured medical evaluation can help clarify what is happening inside the body.
In clinical longevity practice, including the work we do at Soma Wellness Clinic, the first step is understanding how these biological systems are functioning before recommending any supportive strategies.
Longevity medicine, therefore, focuses on something deeper than appearance or isolated symptoms. The goal is to understand how different biological systems age over time and how they can be supported to maintain long-term health and vitality.
Understanding Your Biological Age with Soma Wellness Clinic
One of the most useful steps in longevity care is understanding how the body is functioning at a biological level. Biomarker testing can reveal patterns that are not always visible through symptoms alone.
Markers such as inflammatory indicators, metabolic health measures, hormone levels, and epigenetic age estimation help provide a clearer picture of how the body is ageing internally.
At Soma Wellness Clinic, we build longevity care around physician guided evaluation and personalised monitoring. Our focus is to understand each individual’s biological profile before recommending supportive strategies.
Our longevity programmes may include:
- Biological age and biomarker analysis
- Hormone and metabolic health assessment
- Support for mitochondrial and cellular energy pathways
- Physician supervised longevity therapies
- Personalised guidance on nutrition, sleep and recovery
Through this approach, we aim to support cellular vitality, hormonal balance, and long term resilience. When ageing is examined carefully and monitored over time, it becomes easier to make informed decisions that support both health and quality of life.
Begin your longevity journey with a personalised biological age assessment at Soma Wellness Clinic.
References
1. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The Hallmarks of Aging. Cell. 2013;153(6):1194–1217.
https://www.cell.com/cell/fulltext/S0092-8674(13)00645-4
2. Epel ES, Blackburn EH, Lin J, et al. Accelerated telomere shortening in response to life stress. Proceedings of the National Academy of Sciences of the United States of America (PNAS). 2004;101(49):17312–17315.
https://www.pnas.org/doi/10.1073/pnas.0407162101
3. Horvath S. DNA methylation age of human tissues and cell types. Genome Biology. 2013;14(10):R115.
https://genomebiology.biomedcentral.com/articles/10.1186/gb-2013-14-10-r115
4. Franceschi C, Campisi J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. The Journals of Gerontology: Series A. 2014;69(Suppl_1):S4–S9.
https://academic.oup.com/biomedgerontology/article/69/Suppl_1/S4/561367
5. World Health Organization. International Classification of Diseases 11th Revision (ICD-11): 8E49 Post viral fatigue syndrome.
https://icd.who.int/browse11/l-m/en#/http://id.who.int/icd/entity/569175314
6. Sun N, Youle RJ, Finkel T. The Mitochondrial Basis of Aging. Molecular Cell. 2016;61(5):654–666.
https://www.cell.com/molecular-cell/fulltext/S1097-2765(16)00065-7
7. Snyder PJ, Bhasin S, Cunningham GR, et al. Effects of Testosterone Treatment in Older Men. New England Journal of Medicine. 2016;374:611–624.
https://www.nejm.org/doi/full/10.1056/NEJMoa1506119
8. Jackowska M, Hamer M, Carvalho LA, et al. Short sleep duration is associated with shorter telomere length in healthy men. PLoS ONE. 2012;7(10):e47292.
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0047292
9. Liguori I, Russo G, Curcio F, et al. Oxidative stress, aging, and diseases. Clinical Interventions in Aging. 2018;13:757–772.
https://www.dovepress.com/oxidative-stress-aging-and-diseases-peer-reviewed-fulltext-article-CIA
10. Baker DJ, Childs BG, Durik M, et al. Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature Medicine. 2016;22:1291–1297.
https://www.nature.com/articles/nm.4185
11. Xu M, Pirtskhalava T, Farr JN, et al. Senolytics improve physical function and increase lifespan in old age. Nature Medicine. 2018;24:1246–1256.
https://www.nature.com/articles/s41591-018-0092-9
12. National Institute for Health and Care Excellence (NICE). Menopause: diagnosis and management (NG23). 2015, updated 2019.
https://www.nice.org.uk/guidance/ng23
FAQs
1. What are the signs of premature ageing?
Premature ageing does not usually start with wrinkles. It often begins with minor changes in how you feel. You may experience chronic fatigue, poor sleep, delayed recovery from activity, thinning hair, low mood, decreased muscle strength, or unexpected weight gain. Some people notice early fine wrinkles or lacklustre skin. If several of these changes develop earlier than expected, it could indicate that the body is ageing rapidly on a biological scale.
2. What causes chronic fatigue as we get older?
Feeling a little more fatigued with age is acceptable, but chronic fatigue should not be overlooked. It can be caused by hormonal fluctuations, inadequate sleep, long-term stress, inflammation, low iron, thyroid disorders, or decreased energy synthesis in the body’s cells. If you’re fatigued most days and rest doesn’t seem to help, it’s worth investigating the root problem.
3. Can premature ageing be reversed or slowed down?
Ageing cannot be stopped, but the pace of biological ageing can often be slowed. Healthy habits such as regular exercise, balanced nutrition, good sleep and stress management all play an important role. In some cases, medical support for hormone or metabolic health may also help. When these changes are identified early, it becomes easier to support healthier ageing over time.
The phrase “zombie cells” can sound alarming. It suggests something unnatural or dangerous moving through the body. In reality, the term is simply a nickname used to describe a well recognised biological process.
Zombie cells are scientifically known as Senescent Cells. These are cells that have stopped dividing but continue to remain active within tissues. They are not infectious, they do not multiply uncontrollably and they are not a newly discovered disease.
However, their behaviour has drawn increasing attention from scientists studying ageing and longevity.
Research over the past two decades has shown that senescent cells may influence inflammation, tissue repair and age related decline. Understanding what they are and how they function is becoming an important part of modern longevity science.
What Is a Senescent Cell?
Every cell in the body experiences a life cycle. Apoptosis is a controlled biological process that causes cells to grow, perform specialised functions and eventually die.
This continual cycle of renewal helps tissues to retain their natural form and function.
However, stress can cause a cell to stop this cycle. DNA damage, oxidative stress, repeated replication, or environmental exposure can all cause a defensive response known as Cellular Senescence.
Senescence is the irreversible cessation of cell division.
This procedure is not a mistake. In reality, it has a significant defensive function. Senescence helps to lower the risk of cancer and uncontrolled cell proliferation by preventing damaged cells from replicating.
The trouble emerges when these cells collect rather than being eliminated from the body.
Why Senescent Cells Accumulate With Age
In younger people, the immune system is usually capable of recognising and eliminating senescent cells efficiently. As people age, this cleansing procedure becomes less effective. As a result, senescent cells eventually accumulate in many tissues throughout the body.
These cells continue to be metabolically active, releasing a range of signalling chemicals such as inflammatory proteins, enzymes and growth factors. Scientists refer to this behaviour as the Senescence-Associated Secretory Phenotype, or SASP.
SASP signalling can affect adjacent cells and change the tissue environment. When present in significant numbers, senescent cells can contribute to a condition of persistent low-level inflammation known as Inflammaging. This chronic inflammatory milieu has been connected to a number of age-related biological processes.
According to research published in magazines such as Nature and Nature Medicine, senescent cell accumulation can affect tissue degradation, diminished regenerative ability and metabolic decline.
A widely regarded study undertaken by Mayo Clinic scientists found that selectively eliminating senescent cells from laboratory mice prevented the development of various age-related diseases while improving physical function. Although human research is still ongoing, these findings have helped to establish cellular senescence as one of the key features of ageing biology.
Effects on Tissues and Skin
Senescent cells can appear in many tissues including muscle, blood vessels, organs and skin.
In healthy skin, fibroblast cells produce collagen and elastin that maintain structural strength and elasticity. With age, some fibroblasts enter senescence.
When this occurs, collagen production decreases while inflammatory signalling increases. Over time this can weaken the dermal matrix, contributing to thinning skin, reduced elasticity and the formation of wrinkles.
These changes illustrate an important point. Ageing is not only a surface level process. It is influenced by cellular behaviour deep within tissues.
This is why modern longevity research increasingly focuses on biological mechanisms rather than only cosmetic treatments.
Can Senescent Cells Be Targeted?
Scientists are looking into ways to minimise the load of senescent cells in tissues as they gain a better knowledge of the phenomenon.
Senolytics are one type of chemical that is being researched. These are drugs that are intended to selectively destroy senescent cells while keeping healthy cells intact.
Experimental investigations have looked at combinations like Dasatinib and Quercetin, as well as naturally occurring compounds like Fisetin.
Animal trials have yielded promising results, including reduced inflammation and improved physical function. Early human trials are also ongoing, although long-term safety and clinical efficacy are still being assessed.
Researchers are also investigating how lifestyle variables affect cellular ageing. Regular exercise, metabolic health and nutritional balance appear to help preserve cellular resilience.
While the science continues to advance, it is widely acknowledged that any therapies targeting senescent cells should be addressed with caution and under adequate medical care.
Longevity From The Inside Out: Our Approach at Soma Wellness Clinic
At Soma Wellness Clinic, we view ageing as a biological process influenced by cellular health, metabolic balance and systemic resilience.
Rather than focusing only on visible symptoms, we look at the underlying biological conditions that influence how the body ages.
Cellular senescence is one of the areas we carefully evaluate.
Senescent cells can accumulate over time and may contribute to inflammatory signalling within tissues. Our goal is to support the body in maintaining healthier cellular environments through medically guided longevity strategies.
How We Support Cellular Vitality
Our approach begins with careful clinical evaluation. We conduct biomarker based senescence testing under physician guidance to better understand an individual’s biological ageing profile. This allows us to identify patterns related to inflammation, metabolic health and cellular stress.
Where clinically appropriate, we may explore physician supervised senolytic protocols designed to support the body’s ability to manage senescent cell burden.
In addition, we investigate strategies that support mitochondrial function and cellular energy production. These may include peptide therapies and mitochondrial restoration approaches provided under medical supervision.
Lifestyle also plays a critical role in longevity. Our programmes incorporate detoxification strategies and guidance aimed at reducing chronic inflammation, supporting the body’s natural repair systems.
What Clients Often Experience
Clients participating in our longevity programmes often report improvements in energy levels, recovery and overall vitality.
Individual results vary depending on health status and biological factors. Our focus remains on supporting the body’s ability to restore balance at the cellular level through careful, science based care.
Healthy ageing begins within the body’s cells. By addressing biological ageing mechanisms directly, it becomes possible to support resilience, function and wellbeing over the long term.
References
- What is Cellular Senescence
- Childs BG et al. Cellular senescence in ageing and age related disease. Nature Medicine. 2015.
https://www.nature.com/articles/nm.4000
- Kirkland JL, Tchkonia T. Senolytic drugs from discovery to translation. Journal of Internal Medicine. 2020.
https://onlinelibrary.wiley.com/doi/10.1111/joim.13141
- Justice JN et al. Senolytics in idiopathic pulmonary fibrosis. EBioMedicine. 2019.
https://pubmed.ncbi.nlm.nih.gov/30616998
FAQs
1. What are zombie cells in the body?
Zombie cells are ageing cells that have stopped dividing but do not die when they should. Their scientific name is senescent cells. They stay inside the body and release inflammatory signals that can affect nearby healthy cells. Over time, if too many of them build up, they may contribute to ageing and tissue damage.
2. Are senescent cells bad?
Not always. In fact, senescent cells can be helpful at first. They stop damaged cells from turning into cancer. The problem happens when too many of them collect in the body as we age. When they build up, they can increase inflammation and slow down tissue repair. It is the accumulation that causes concern, not the process itself.
3. Can you remove zombie cells naturally?
Although research is ongoing, certain healthy lifestyle behaviours may help your body better handle senescent cells. Regular exercise, adequate sleep, balanced nutrition and a healthy weight may assist the immune system eliminate damaged cells. Scientists are also exploring chemicals known as senolytics, which should only be used under medical supervision.
4. Do zombie cells cause wrinkles and skin ageing?
Senescent cells can damage the skin. As skin cells age, some become senescent and cease to produce adequate collagen. They may also produce inflammatory substances, which degrade the skin structure. This can lead to wrinkles and a loss of firmness with time. However, skin ageing is influenced by a variety of factors, including sun exposure, genetics and lifestyle.
Chronic inflammation is at the root of many modern health conditions, including arthritis, heart disease and autoimmune illnesses. It discreetly promotes tissue deterioration, weariness and discomfort, often defying standard treatments. Enter EBOO Therapy or Extracorporeal Blood Oxygenation and Ozonation, a cutting-edge technique gaining popularity for its ability to tackle inflammation at its source. But does science support its claims? This blog explains what EBOO is, how it differs from typical ozone therapy and looks at the data for its role in reducing chronic inflammation.
What Is EBOO Therapy?
EBOO Therapy is a significant advancement in ozone-based treatments. It operates by drawing up to 4.8 litres of blood from the body through an intravenous line. This blood is then sent via a specialist machine that resembles dialysis equipment. It goes through filtration to remove poisons, germs and inflammatory material. Medical-grade ozone gas, mixed with pure oxygen, is pumped into the blood at precise concentrations, usually 0.5 to 1 microgram per millilitre. The procedure oxygenates red blood cells, boosts antioxidant synthesis and allows pure blood to circulate uninterrupted. In simple layman term, blood is continuously withdrawn and returned in a closed-loop system.
A normal session lasts 45-60 minutes. Although outcomes vary, certain individuals begin experiencing positive shifts in vitality and mood within the first one to two sessions. EBOO, developed in Europe in the early 2000s, is based on ozone therapy principles but is significantly scaled up. Unlike simple injections, it uses a complete blood volume equivalent to achieve systemic cleansing and immunological recalibration.
EBOO Vs Traditional Ozone Therapy
Ozone therapy dates back to the 1930s and is used for disinfection and mild autohemotherapy, in which only 250 millilitres of blood are taken, ozonated and reinjected. Rectal insufflation, joint injections and IV drips are all possible methods. These provide ozone locally or in small dosages, making them ideal for treating acute conditions such as infections or wounds.
EBOO distinguishes out for its ability to handle continuous, high-volume blood flow. In some forms, ozonation is combined with haemofiltration to better filter out microclots, lipids and cytokines. Proponents say that this enhances ozone’s biphasic action: initial oxidative stress causes a rebound in antioxidants such as glutathione and superoxide dismutase, which modulates immunity.
The Science Behind EBOO’s Anti-Inflammatory Effects
To understand how EBOO may affect inflammation, we must examine how ozone interacts with blood on a biological level.
Medical ozone (O₃) at controlled quantities reacts rapidly with plasma lipids and antioxidants. This reaction produces short-lived compounds called ozonides and lipid oxidation products. When delivered in therapeutic doses, these compounds function as biological signalling messengers rather than poisons.
One of the most important routes active is the Nrf2 (Nuclear factor erythroid 2-related factor 2) pathway. Nrf2 is a transcription factor that controls the production of endogenous antioxidants including glutathione, catalase and superoxide dismutase. Nrf2 activation aids the body’s defence against oxidative stress, which is intimately related to chronic inflammation.
At the same time, controlled oxidative signaling may help modulate pro-inflammatory cytokines such as:
- IL-6 (Interleukin-6)
- TNF-α (Tumor Necrosis Factor-alpha)
- CRP (C-reactive protein)
These markers are often increased in chronic inflammatory conditions.
A 2022 review of oxygen-ozone therapy published on PubMed Central, “Oxygen-Ozone Therapy for Reducing Pro-Inflammatory Cytokines” (PMC8910188), examined how ozone therapy can reduce inflammatory cytokines while increasing antioxidant capacity.
There is also evidence that ozone therapy may improve endothelial function, or the health and responsiveness of the blood vessel’s inner lining. Endothelial dysfunction is a crucial factor in cardiovascular inflammation and atherosclerosis.
Furthermore, these pathways are dose-dependent. Ozone, at controlled medicinal concentrations, functions as a regulated oxidative stimulation rather than causing uncontrolled oxidative damage.
Clinical Evidence: Strengths and Studies
The landmark study remains a 2005 randomised controlled trial by Di Paolo et al., involving 28 patients with peripheral artery disease (PAD). Participants received either EBOO (three sessions weekly for seven weeks) or prostacyclin infusions. EBOO excelled: skin lesions regressed significantly (p<0.001), pain, pruritus and leg heaviness eased markedly and subjective well-being soared. No vascular changes occurred and 210 total sessions proved safe.
Earlier safety trials (2000-2005) treated more than 82 patients with PAD, cardiac problems and hepatitis C, proving tolerance at therapeutic levels with no harm. Bocci’s 2005 assessment emphasised EBOO’s biological plausibility, which activates immunological and endothelial cells through regulated oxidation.
A meta-analysis from Aspen Medical Centre (12 studies, 2020-2024; 1,247 patients) found 67% energy gains after one to three sessions and 85% total improvement after the program. Related ozone studies, such as a 2022 study of pro-inflammatory cytokines, suggests improvements in osteoarthritis and low back pain.
Practical Applications and Patient Outcomes
Clinics offering EBOO report use in patients experiencing:
- Chronic fatigue syndromes
- Fibromyalgia
- Autoimmune-related inflammation
- Post-viral recovery
- Persistent inflammatory states
Some patients report improvements in:
- Energy levels
- Mental clarity
- Joint discomfort
- Exercise tolerance
Because EBOO processes a large blood volume, proponents believe it offers a more systemic reset compared to small-volume ozone therapies.
However, outcomes vary depending on underlying condition, metabolic health and overall treatment strategy.
EBOO is typically used as part of a broader plan that may include nutritional therapy, lifestyle optimisation and conventional medical care where necessary.
Is EBOO Worth Considering? And How We Use It at SOMA Wellness
Chronic inflammation is rarely caused by one single factor. It builds slowly, influenced by stress, metabolism, environment and immune signaling. Because of that, treatment often requires more than a single pill.
EBOO is being explored as part of that broader approach. The current research base is still growing, but early clinical studies and physiological data suggest it may help support inflammatory regulation and oxidative balance in selected patients. It is best viewed as supportive, not standalone.
At SOMA Wellness, we integrate EBOO only after careful medical evaluation. We use it as part of a structured recovery strategy, especially for individuals experiencing persistent fatigue, inflammatory overload or prolonged recovery.
- Our approach focuses on:
- Restoring cellular energy efficiency
- Supporting balanced immune response
- Improving oxygen utilisation
- Strengthening systemic resilience
We believe that innovative medicines deserve enhanced oversight. Certain protocols are only supplied following a physician’s evaluation and when clinically suitable.
For the right person, at the right time, EBOO can be a significant step in regaining momentum in health. As science advances, so does our ability to use it more precisely and responsibly.
References
- Di Paolo N, et al. (2005). Extracorporeal blood oxygenation and ozonation (EBOO): a controlled trial…
https://pubmed.ncbi.nlm.nih.gov/16288443/
- Bocci V., et al. (2005). Extracorporeal blood oxygenation and ozonation: clinical and biological implications…
https://pubmed.ncbi.nlm.nih.gov/16156950/
- Oxygen-ozone therapy review (2022). Effects on pro-inflammatory cytokines.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8910188/
- Case report of toxin reduction post-EBOO (2025).
https://pmc.ncbi.nlm.nih.gov/articles/PMC12826612/
FAQs
1. Is EBOO therapy scientifically proven?
Early clinical studies on EBOO therapy have demonstrated a strong safety profile and promising biological effects, particularly in vascular and inflammatory conditions. While many of the current studies are small, the growing body of research continues to build encouraging momentum, with larger randomised controlled trials anticipated as interest expands. EBOO is increasingly being recognised as a valuable integrative therapy that complements conventional treatments and supports overall vascular and systemic health.
2. How is EBOO different from regular ozone therapy?
Traditional ozone therapy usually treats a small amount of blood or delivers ozone locally. EBOO processes a much larger blood volume in a continuous, controlled system. Because of this, it is considered a more intensive and systemic form of ozone therapy.
3. Can EBOO therapy reduce chronic inflammation?
Studies suggest that ozone therapy may help manage inflammatory indicators including CRP, IL-6 and TNF-alpha by stimulating the body’s antioxidant mechanisms. While first findings are promising, the outcome varies depending on the individual and underlying health condition.
4. Is EBOO therapy safe?
When performed under appropriate medical supervision and established clinical protocols, EBOO has demonstrated a favourable safety profile in early studies. It is not suitable for every individual and requires proper medical screening. At SOMA Wellness, all patients undergo a physician-led evaluation before treatment and the procedure is conducted in a controlled clinical setting with continuous monitoring to ensure safety and suitability.
In the relentless quest for weight loss, society has often overlooked the most vital aspects of true health and well-being. The narrative around weight loss has long been dominated by simplistic equations—calories in versus calories out—and narrow ideals that prize thinness above true metabolic integrity. As a result, we have reduced our health aspirations to chasing ever-smaller numbers on a scale, believing that a shrinking body automatically equates to a healthier one. Scales become the ultimate judges of success, calories are demonized, and bodies are measured solely in terms of appearance rather than function, resilience, or vitality.
But beneath this cultural obsession lies a deeper and more revolutionary truth—one that challenges the conventional paradigm. Real, lasting, and meaningful weight management is not about deprivation; it’s about preservation. It’s about holding onto what matters most—muscle, bone, mitochondrial health, hormonal harmony, and cognitive clarity—while shedding excess fat and inflammation. It’s about cultivating a system that’s robust and vibrant, not simply lighter. This shift in mindset reframes weight loss as a process of gaining strength, longevity, and inner stability—not just losing weight for aesthetics or short-term gratification.
Far too many people have embarked on weight loss journeys only to end up depleted—physically, emotionally, and metabolically. We must ask ourselves: are we pursuing health, or simply thinness? Sustainable weight management is about optimizing every system in the body, not starving them. The real prize isn’t dropping sizes—it’s gaining strength, resilience, and an enduring quality of life.
Lean Mass: The Metabolic Gold
Muscle is not a luxury; it’s a necessity. In the evolving conversation around weight loss, preserving lean body mass—particularly skeletal muscle—has emerged as a critical yet often neglected priority. Beyond aesthetics or performance, lean mass is fundamentally tied to overall metabolic health, insulin sensitivity, immune resilience, and even mental vitality. Muscle tissue is a metabolically active organ that not only consumes energy at rest but also acts as a powerful regulator of systemic glucose levels, inflammation, and hormonal balance. As we age, the gradual loss of muscle mass—known as sarcopenia—can quietly shift the balance from healthy weight loss to a cascade of health challenges including insulin resistance, frailty, increased fall risk, and accelerated biological aging [1].
When people pursue aggressive calorie restriction or over-exercise without strategic support, the body often breaks down muscle to meet its energy demands. This muscle loss weakens not just physical strength but the very foundation of metabolic health. Lean muscle functions as a reservoir for glucose and amino acids, providing both energy and essential substrates during times of stress or caloric deficit [2]. Just as importantly, muscle acts as an endocrine organ, releasing beneficial signaling molecules called myokines during contraction. These myokines exert anti-inflammatory, anti-diabetic, and even anti-tumor effects, helping the body maintain homeostasis across systems—from the brain to the liver and immune system [3]. Thus, muscle isn’t just something to “tone up”—it’s an essential, active player in the body’s weight regulation machinery.
To protect muscle during weight loss, several interventions must work in tandem. First and foremost is resistance training, ideally performed at least three to four times per week. Compound movements like squats, deadlifts, and push-pull sequences activate large muscle groups and stimulate protein synthesis, which not only prevents loss but may build new muscle even in a calorie deficit. Second, adequate protein intake is crucial—aiming for 1.2 to 2.0 grams of protein per kilogram of body weight per day is a clinically supported benchmark [4]. This becomes especially important with age, when anabolic resistance increases the protein requirements for maintaining muscle tissue. Supplementation with branched-chain amino acids (BCAAs), particularly leucine, can help preserve lean mass during weight loss and intermittent fasting, providing targeted stimulation of mTOR for muscle synthesis without excessive overall calories [5].
Beyond protein and training, strategic support with nutrients like magnesium citrate can improve muscular recovery and reduce post-exercise inflammation.
Similarly, riboflavin (B2) and niacin (B3), taken in higher therapeutic doses, can enhance mitochondrial efficiency within muscle cells—optimizing ATP production, reducing oxidative stress, and boosting endurance capacity. For individuals pursuing advanced mitochondrial support, compounds like methylene blue are emerging as adjuncts that enhance electron transport and reduce cellular fatigue, potentially preserving muscle function during caloric restriction.
Resveratrol, a polyphenol found in red grapes and berries, also supports lean mass by activating the SIRT1-AMPK pathway, which indirectly inhibits mTOR overactivation while enhancing mitochondrial biogenesis and insulin sensitivity. Its mild mTOR modulation may allow for fat loss while preserving muscle, especially in those undergoing intermittent fasting or ketogenic nutrition. Moreover, resveratrol’s anti-inflammatory and antioxidant properties help blunt catabolic signals that can otherwise accelerate muscle breakdown in calorie-deficit states.
Another crucial yet underappreciated tool in maintaining lean mass during weight management is pregnenolone, the master neurosteroid and precursor to hormones like DHEA, progesterone, testosterone, and cortisol. During extended stress, fasting, or caloric restriction, the body may shunt hormone production toward cortisol at the expense of anabolic and neuroprotective pathways—a process known as pregnenolone steal. Supplementing with low physiological doses of pregnenolone may help buffer against this depletion, promoting more balanced endocrine signaling. This can support muscle protein synthesis indirectly by stabilizing testosterone and DHEA levels, while also improving cognitive clarity and motivation—factors often compromised during prolonged dieting. Pregnenolone’s ability to modulate GABA and NMDA receptors also contributes to better sleep and recovery, both of which are essential for muscle preservation and repair.
Bone Density: The Silent Strength
Bone may seem inert and structural, but in truth, it is a dynamic, living tissue that communicates constantly with the rest of the body. In the context of weight loss, bone health is often an afterthought—but it shouldn’t be. Weight loss that leads to the erosion of bone mass can quietly accelerate biological aging, especially in vulnerable populations such as postmenopausal women, individuals on restrictive diets, and the elderly [6]. The consequences aren’t merely cosmetic or mobility-related; reduced bone density increases the risk of fractures, chronic pain, and long-term disability. Moreover, it often happens silently, without overt symptoms, until a major injury reveals the underlying fragility.
Emerging science shows that the relationship between bone and metabolism is far more intimate than previously understood. Bone is not just scaffolding—it’s a metabolically active endocrine organ. Hormones like osteocalcin, secreted by bone-forming osteoblasts, have systemic effects, including enhancing insulin sensitivity, increasing testosterone production, and improving mitochondrial function [7]. This means that bone health is intertwined with the very processes we seek to optimize during weight loss: hormonal balance, energy production, and metabolic control. Compromising bone in the pursuit of fat loss may paradoxically impair the body’s ability to maintain that loss.
To preserve and build bone during weight loss, several strategies should be prioritized. Weight-bearing exercises—especially resistance training, yoga, hiking, and bodyweight movements—stimulate the mechanical loading needed to trigger bone remodeling. Unlike cardio alone, resistance exercises apply specific stress to bones, prompting adaptive strengthening and improved density over time. This is essential not only for maintaining skeletal strength but also for enhancing joint integrity and posture.
Nutritional support is equally vital. Vitamin D3 enhances calcium absorption, while vitamin K2 helps shuttle calcium into bones and away from arteries—an often-overlooked nuance in preventing vascular calcification. Among minerals, magnesium citrate stands out due to its superior bioavailability and dual action: it supports both bone mineralization and muscle relaxation, making it invaluable during weight loss regimens that include increased physical training [8]. Magnesium is also involved in over 300 enzymatic processes, many of which influence bone turnover, hormonal balance, and inflammation regulation.
Tracking progress through periodic DEXA scans is a smart and often underutilized strategy [9]. DEXA not only provides a snapshot of bone density but also differentiates between fat mass and lean mass, offering a more complete picture of body composition. For those serious about sustainable, health-promoting weight loss, knowing what’s being lost—fat, muscle, or bone—can be the difference between transformation and regression. Prioritizing bone health isn’t just about preventing fractures; it’s about safeguarding the silent strength that upholds metabolic vitality, mobility, and longevity.
The Role of Inflammation
Fat is not merely passive storage for excess calories—it is a hormonally active, immunologically significant organ system that communicates continuously with the rest of the body. In particular, visceral fat, the type that surrounds internal organs, acts as an endocrine organ that secretes a range of pro-inflammatory cytokines, including TNF-α (tumor necrosis factor alpha) and IL-6 (interleukin-6). These inflammatory mediators interfere with insulin receptor signaling, promoting systemic insulin resistance, dysregulated glucose metabolism, and chronic low-grade inflammation—hallmarks of metabolic syndrome and type 2 diabetes [10].
This persistent inflammatory milieu does more than just stall weight loss efforts; it actively degrades health on multiple fronts. Chronic inflammation damages the endothelial lining of blood vessels, accelerating the development of atherosclerosis and increasing cardiovascular risk. It also contributes to neuroinflammation, impairing cognitive performance, mood, and increasing the risk of neurodegenerative diseases such as Alzheimer’s. On a cellular level, inflammation directly impairs mitochondrial function, decreasing energy production, increasing reactive oxygen species (ROS), and promoting fatigue, aging, and metabolic inefficiency. Thus, inflammation is not just a symptom but a central driver of disease and weight regulation failure.
The solution lies in comprehensive inflammation control—not through suppressing the immune system, but by retraining and nourishing it through strategic interventions. A whole-food, anti-inflammatory diet is foundational. This includes generous intake of polyphenol-rich fruits and vegetables, fiber to feed beneficial gut bacteria, and omega-3 fatty acids from sources like walnuts, and fatty fish [11]. These nutrients help reduce the production of pro-inflammatory mediators and increase anti-inflammatory counterparts like IL-10 and adiponectin.
Beyond nutrition, lifestyle strategies such as mindful stress reduction, high-quality sleep, and intermittent fasting help rebalance the hypothalamic-pituitary-adrenal (HPA) axis and reduce cortisol-mediated inflammation [12]. Even moderate exercise can lower C-reactive protein (CRP) levels and stimulate anti-inflammatory myokines from skeletal muscle. These habits, when practiced consistently, form the foundation of an anti-inflammatory lifestyle.
Targeted supplementation provides another layer of support. Curcumin, the active compound in turmeric, inhibits the NF‑κB pathway, a master regulator of inflammation. Ginger, with its natural COX-2-inhibiting properties, also offers synergistic anti-inflammatory action. Of special interest is resveratrol, a plant-derived polyphenol that activates sirtuins, particularly SIRT1, which play a critical role in modulating inflammatory responses and mitochondrial function [13]. Resveratrol not only blunts the cytokine storm associated with visceral fat but also helps recalibrate the body’s inflammatory thermostat at a molecular level. Its dual role in mitochondrial protection and inflammatory modulation makes it an ideal candidate for individuals looking to reduce fat mass without compromising metabolic resilience.
By recognizing and targeting inflammation as a root cause—not just a side effect—of weight challenges, we lay the groundwork for more sustainable and meaningful health outcomes. Inflammation is not just a fire to be put out; it is a biological signal that, when properly interpreted and addressed, can guide us toward deeper healing, improved vitality, and longer-lasting weight regulation.
Liver Health: The Metabolic Command Center
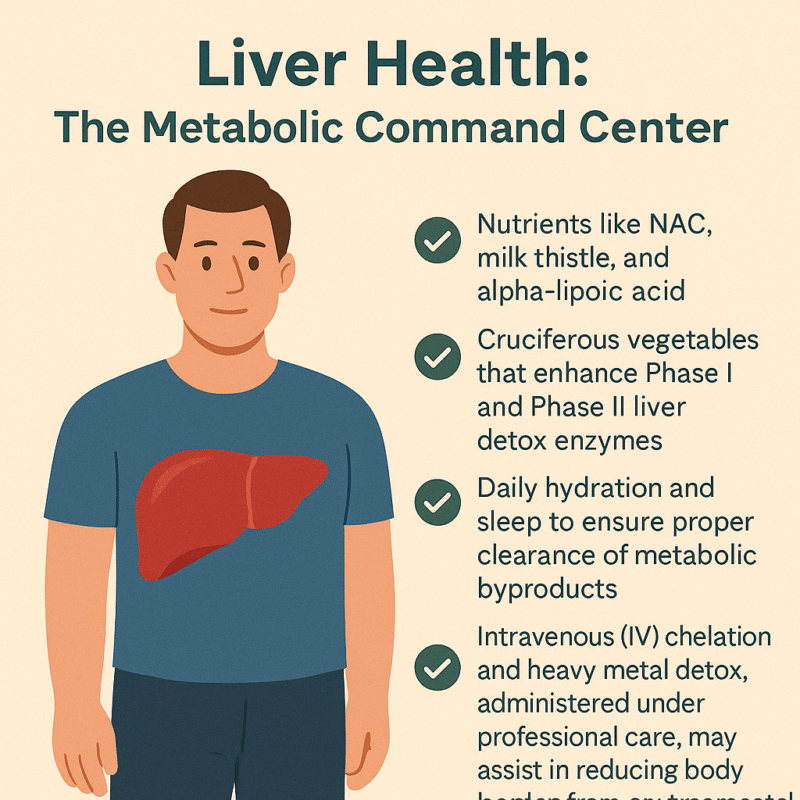
Often overlooked in weight loss conversations, the liver plays a truly central role in regulating metabolism, hormone balance, detoxification, and fat processing. It is the body’s metabolic command center, coordinating an astonishing number of chemical reactions every day—more than 500 in total. From metabolizing carbohydrates and breaking down fats to neutralizing toxins and converting hormones, the liver is integral to maintaining internal balance. When the liver is sluggish or overburdened—due to poor diet, excessive alcohol, prescription drugs, or environmental toxins—its ability to process fats and regulate glucose declines. This dysfunction can create a metabolic bottleneck, making weight loss difficult, even in people who are otherwise eating well and exercising.
A compromised liver may allow fatty deposits to accumulate within its own tissue, a condition known as non-alcoholic fatty liver disease (NAFLD). NAFLD is now one of the most common liver conditions worldwide and is strongly associated with insulin resistance, metabolic syndrome, and central obesity [14]. As the liver struggles to detoxify and process nutrients efficiently, inflammation and oxidative stress can build up systemically, impairing not just fat loss but also energy levels, immune function, and hormonal health. Since the liver also helps metabolize estrogens and other steroid hormones, a sluggish liver can worsen estrogen dominance or hormonal imbalance, especially during perimenopause and andropause.
Supporting liver function is thus not just helpful—it is essential for healthy, sustainable weight management. A multipronged approach is best. Certain nutraceuticals are well-researched for their liver-protective properties. N-acetyl cysteine (NAC) boosts intracellular glutathione, the body’s master antioxidant, helping neutralize free radicals and support Phase II detoxification. Milk thistle (silymarin) has been shown to stabilize liver cell membranes and stimulate protein synthesis in hepatocytes, while alpha-lipoic acid supports both liver regeneration and insulin sensitivity [14]. These compounds create a protective biochemical environment that enhances fat metabolism and reduces oxidative damage.
Diet also plays a major role in liver health. Cruciferous vegetables—like broccoli, cauliflower, Brussels sprouts, and kale—are rich in glucosinolates, which stimulate Phase I and Phase II detox enzymes in the liver. These enzymes help convert lipophilic toxins into water-soluble forms that can be safely excreted [15]. The fiber in these vegetables also supports gut-liver axis function by binding toxins and facilitating regular elimination. Combined with sufficient hydration and deep, restorative sleep, these practices help ensure that the liver can efficiently clear metabolic byproducts and prevent the recirculation of toxins in bile.
In more complex or toxin-burdened individuals, intravenous (IV) detoxification strategies may be warranted. Chelation therapy, using agents like EDTA or N acetyl cycteine, under medical supervision, can assist in binding and removing heavy metals such as lead, mercury, and cadmium—substances known to impair mitochondrial function and enzyme activity in the liver. These toxins, when left unchecked, contribute not only to fatty liver and oxidative stress but also to hormonal disruption and sluggish metabolism. Regular assessments and guided detox protocols can significantly reduce this toxic load, often resulting in better energy, improved body composition, and enhanced clarity of mind.
By treating the liver as the metabolic powerhouse that it is, and not merely a silent filter, we create the internal conditions for real transformation. A well-functioning liver clears the path for hormone balance, inflammation reduction, effective fat burning, and ultimately, sustainable weight loss from the inside out.
Brain and Joint Health: Weight Loss Without the Cost
In the pursuit of rapid weight loss, it’s easy to focus exclusively on calories, exercise, and appearance—yet often forgotten are the two pillars of long-term vitality and quality of life: the brain and the joints. Unfortunately, overly aggressive caloric restriction or nutrient-poor diets can have unintended consequences that affect these critical systems. When the body is deprived of key macronutrients and micronutrients, it may cannibalize structural proteins, reduce neurotransmitter synthesis, and accelerate cartilage breakdown. The result? Cognitive fog, mood instability, and increased joint pain—symptoms that are not only discouraging but can derail even the most well-intentioned weight management plans.
The brain is metabolically demanding, consuming over 20% of the body’s energy at rest. It depends on a continuous supply of healthy fats, particularly long-chain omega-3 fatty acids (EPA and DHA), stable glucose levels, and micronutrients like B vitamins, magnesium, and antioxidants to maintain its functions—memory, mood regulation, focus, and neuroplasticity. Erratic blood sugar levels, inflammation, and oxidative stress can impair synaptic efficiency and neurotransmitter production, contributing to fatigue, depression, and anxiety. Similarly, joints require structural proteins such as collagen, as well as synovial fluid support from compounds like hyaluronic acid and methylsulfonylmethane (MSM) to stay resilient under mechanical load, especially during weight-bearing exercises.
To support both neurological and musculoskeletal systems during weight loss, a foundational approach includes ensuring sufficient intake of omega-3 fatty acids, particularly EPA and DHA, which are vital for neuronal membrane fluidity and the regulation of inflammation in both the brain and joints [16]. These fatty acids not only protect against neurodegeneration but have been shown in studies to improve mood, reduce anxiety, and support executive function. For those with dietary restrictions, high-quality fish oil or algae-based omega-3 supplements can provide these benefits with minimal caloric burden.
Incorporating low-impact, joint-friendly movement—such as swimming, yoga, Pilates, or brisk walking—provides the dual benefit of stimulating circulation and maintaining mobility without excessive mechanical strain. This movement also supports lymphatic drainage, which is essential for clearing inflammatory metabolites that can accumulate with tissue breakdown or detoxification.
One underappreciated but powerful ally in preserving both cognitive clarity and joint integrity is pregnenolone, a neurosteroid produced in the brain and adrenal glands. As the precursor to key hormones like DHEA, progesterone, testosterone, and cortisol, pregnenolone plays a central role in neuroendocrine balance. It modulates receptors such as GABA and NMDA, contributing to enhanced mood stability, reduced anxiety, and sharper mental performance. Additionally, some emerging research suggests that pregnenolone may influence connective tissue metabolism and joint resilience, potentially improving recovery and reducing pain perception during periods of weight loss and increased physical demand. By supporting neurohormonal homeostasis, pregnenolone can be a valuable tool for avoiding burnout and breakdown while pursuing body recomposition.
When weight loss is approached through a lens that honors the interconnectedness of brain and joint health, the result is a more sustainable, energizing, and empowering journey. Rather than sacrificing cognitive vitality and physical freedom, we can build a path to health that enhances both—allowing us to think, move, and feel better at every stage of transformation.
Mitochondria: Engines of Life
At the cellular level, weight management is mitochondrial management. These tiny, double-membraned organelles serve as the energy factories of our cells, converting nutrients into usable energy in the form of ATP through oxidative phosphorylation. Their efficiency—or lack thereof—directly dictates how well we burn fat, respond to stress, regulate inflammation, and maintain physical stamina. Impaired mitochondrial function doesn’t just lead to fatigue—it lies at the heart of insulin resistance, obesity, neurodegeneration, and accelerated aging [18]. In essence, when mitochondria fail, the whole body slows down—metabolically, cognitively, and immunologically.
Mitochondria do far more than produce energy; they also regulate apoptosis (programmed cell death), modulate the production of reactive oxygen species (ROS), and participate in the cellular stress response. In individuals with metabolic dysfunction, these mitochondria may become swollen, leaky, and inefficient—burning fuel poorly while spilling oxidative byproducts into the surrounding tissue. This “leaky battery” state can lead to a vicious cycle of inflammation, hormonal disruption, and impaired detoxification, all of which sabotage fat loss and degrade overall vitality. A sluggish mitochondrion doesn’t just stall your metabolism; it creates a state of biological inertia.
Optimizing mitochondrial function is therefore a cornerstone of sustainable weight loss, especially for individuals suffering from fatigue, brain fog, or metabolic resistance. One of the most exciting and non-invasive methods for enhancing mitochondrial health is red and near-infrared light therapy. By stimulating cytochrome c oxidase in the mitochondrial electron transport chain, photobiomodulation boosts ATP output, reduces inflammation, and can even trigger mitochondrial biogenesis—the creation of new mitochondria [19]. This makes red light therapy particularly valuable for those with sedentary lifestyles, injuries, or chronic pain who may not yet tolerate vigorous exercise.
Nutritional and nutraceutical interventions also play a powerful role in mitochondrial revitalization. Key compounds such as Coenzyme Q10 (CoQ10), PQQ, and L-carnitine work synergistically to enhance mitochondrial energy output, protect against oxidative damage, and shuttle fatty acids into mitochondria for oxidation. Among these, methylene blue is gaining interest as a novel “electron cycler”—acting as a bypass for damaged segments of the electron transport chain. By accepting and donating electrons, methylene blue improves redox cycling, enhances ATP production, and reduces ROS generation in compromised cells [20]. Used in microdoses, it has shown promise in conditions ranging from mitochondrial myopathy to cognitive decline, and could serve as a valuable tool in resistant weight loss and energy optimization strategies.
High-dose riboflavin (vitamin B2) and niacin (vitamin B3) offer additional mitochondrial support by functioning as precursors to FAD+ and NAD+, respectively—critical cofactors in enzymatic reactions that drive energy metabolism. These nutrients can be particularly helpful in individuals with mitochondrial fatigue, chronic illness, or those exposed to high levels of environmental toxins. Enhancing the NAD+/NADH ratio, a key marker of mitochondrial health, also promotes autophagy and DNA repair, further linking these vitamins to longevity pathways.
An often-underestimated ally in mitochondrial wellness is ascorbic acid (vitamin C). While best known as an antioxidant, vitamin C also plays a cofactor role in carnitine synthesis, collagen production (crucial for mitochondrial membrane integrity), and several steps within the TCA cycle, the energy-producing hub of the mitochondrion. Its antioxidant properties help buffer against mitochondrial-generated ROS, while also supporting adrenal function during periods of metabolic stress.
Lifestyle strategies round out the mitochondrial toolbox. Cold exposure, intermittent fasting, and high-intensity exercise all stimulate mitochondrial biogenesis and autophagy, creating a cellular environment optimized for fat burning and longevity [21]. Fasting in particular activates AMPK and PGC-1α, key regulators of energy balance and mitochondrial renewal, while simultaneously reducing insulin and inflammation.
When mitochondria thrive, the body gains the capacity to lose fat without losing energy, to detoxify without exhaustion, and to age without decline. By nourishing these intracellular engines, we ignite a foundation not only for sustained weight management, but also for vibrant, whole-body health.
Hormones: Conductors of Weight and Wellness
Hormones are the body’s master messengers, orchestrating nearly every physiological process, from metabolism and mood to sleep, libido, and fat storage. In the context of weight management, hormonal balance is not optional—it is foundational. Hormones act like conductors of a symphony: when in harmony, the body hums along with vitality and metabolic clarity; when imbalanced, even the best efforts in nutrition and exercise may falter. Key players like thyroid hormones, cortisol, insulin, estrogen, and testosterone form a dynamic web. A disturbance in one arm of this network often causes ripple effects across the entire system, creating a cascade of dysfunction that leads to stubborn fat gain, fatigue, mood swings, and muscle loss.
The thyroid gland, for instance, regulates basal metabolic rate through hormones T3 and T4. Suboptimal thyroid function—whether due to nutrient deficiencies (like iodine or selenium), autoimmunity, or stress—can significantly reduce calorie burn and slow fat oxidation, even when diet and activity levels are on point [22]. Without proper thyroid signaling, individuals often experience cold intolerance, constipation, and weight gain despite caloric restriction. Supporting thyroid health requires a blend of nutritional sufficiency, gut health, and in some cases, fine-tuning of T3/T4 balance through medical therapy.
Cortisol, the stress hormone, is equally influential. While short-term cortisol spikes can help mobilize energy during stress or fasting, chronic elevation due to unresolved psychological stress, poor sleep, or overtraining can promote abdominal fat storage, suppress thyroid function, and worsen insulin resistance. Modulating cortisol naturally through lifestyle adjustments—such as breathwork, circadian rhythm alignment, and emotional regulation—is powerful. The inclusion of adaptogenic herbs like ashwagandha and Rhodiola rosea has shown to buffer the physiological effects of chronic stress, enhancing resilience while supporting adrenal recovery [23].
Sex hormones such as testosterone and estrogen also play vital roles. In both men and women, low testosterone is associated with increased fat mass and decreased lean muscle. Estrogen, while often vilified, is essential for bone density, glucose regulation, and mitochondrial function. Hormonal imbalances during menopause, andropause, or due to environmental xenoestrogens can shift body composition unfavorably. In cases of clinically confirmed deficiency or dysfunction, bioidentical hormone replacement therapy (BHRT)—tailored to individual needs and monitored closely—can restore physiological balance, improve metabolic flexibility, and enhance quality of life [24].
Ultimately, hormonal optimization is not about manipulating a single hormone in isolation. It’s about understanding the interdependence of systems—the thyroid talking to the adrenals, the pancreas influencing the ovaries, and so forth. A functional medicine approach uses labs, symptom tracking, and lifestyle factors to address root causes and restore rhythm to the body’s hormonal symphony.
Detoxification: Your Weight Loss Accelerator
In today’s world, toxic burden is a hidden variable in many cases of resistant weight loss. From pesticides and plastics to heavy metals and hormone-disrupting chemicals, we are constantly exposed to substances that the body must metabolize and clear. Many of these toxins are lipophilic, meaning they are stored in fat tissue. As the body burns fat during weight loss, these toxins are released back into circulation. Without adequate support, this process can overwhelm detoxification pathways, resulting in fatigue, brain fog, stalled progress, or even hormone disruption.
Detoxification is not just a cleanse—it’s a biochemical process, primarily performed by the liver, gut, kidneys, skin, and lymphatic system. The liver’s Phase I and Phase II detox enzymes transform fat-soluble toxins into water-soluble metabolites that can be excreted through bile or urine. However, this process requires sufficient cofactors such as sulforaphane (from broccoli sprouts), glutathione, B vitamins, and amino acids like glycine and cysteine [25]. If these nutrients are lacking—or if Phase II is sluggish while Phase I is overactive—intermediary toxins can accumulate, increasing oxidative stress and inflammation.
Supporting detox is a layered endeavor. Consuming cruciferous vegetables, berries, and high-fiber foods provides both enzymatic activation and toxin-binding capacity in the gut. Hydration and adequate bile flow (supported by bitters or phosphatidylcholine) are essential for flushing waste through the liver-gallbladder-intestinal axis. Moreover, regular infrared sauna therapy can augment toxin clearance through the skin, while simultaneously stimulating heat shock proteins that protect cellular integrity [26].
The gut, often referred to as the “second liver,” plays a key role in toxin recycling. A compromised intestinal lining can allow enterohepatic recirculation—the reabsorption of bile-bound toxins—further burdening the liver. Maintaining gut integrity with probiotics, immunoglobulins, and fiber reduces this loop and ensures toxins are carried out effectively [27]. Additionally, movement practices such as rebounding, lymphatic massage, and walking support the drainage systems that move cellular waste out of the body.
For individuals with elevated toxic exposure—such as mold, mercury, lead, or persistent organic pollutants—deeper interventions may be required. IV chelation, phospholipid exchange, and high-dose antioxidant IV therapy can accelerate removal of these compounds when performed under professional care. These therapies help to restore mitochondrial and hormonal function by removing the underlying biochemical irritants that interfere with cell signaling and energy production.
Rather than relying on fads or quick cleanses, a structured, nutrient-dense, and practitioner-guided detox protocol aligns with how the body is biologically designed to heal. In the context of weight loss, it allows for the safe release of stored toxins, reducing inflammatory backlash and preserving hormonal and mitochondrial resilience. Done correctly, detoxification becomes not just a side strategy, but a catalyst for deep metabolic renewal.
The EBOO Advantage: Ozone for Metabolism
In the quest to optimize metabolism, reduce inflammation, and support cellular renewal, Extracorporeal Blood Oxygenation and Ozonation (EBOO) has emerged as a promising adjunctive therapy within integrative and functional medicine. This cutting-edge technique involves drawing blood from the patient, infusing it with a carefully calibrated concentration of ozonated oxygen, and reintroducing it into the body in a closed-loop system. The goal is to stimulate a cascade of oxidative preconditioning responses—essentially “training” the body to bolster its antioxidant defenses, optimize circulation, and enhance mitochondrial resilience. Though still considered investigational in many countries, early studies and clinical observations suggest that EBOO may have multi-system benefits, particularly for individuals dealing with metabolic dysfunction, chronic inflammation, or toxic burden [28].
One of the most compelling aspects of EBOO is its impact on systemic inflammation, which lies at the root of obesity, type 2 diabetes, cardiovascular disease, and autoimmune conditions. By modulating cytokine levels, particularly pro-inflammatory markers like IL-6 and TNF-α, EBOO has demonstrated the potential to lower the inflammatory burden associated with metabolic syndrome [29]. In individuals who have plateaued in their weight loss journey or present with persistent fatigue, brain fog, or joint pain—despite lifestyle interventions—EBOO may offer a way to reduce subclinical inflammation and restore metabolic momentum. Its potential to support immune recalibration without immunosuppression makes it uniquely suited for conditions marked by immune overactivity and oxidative stress.
Another key benefit of EBOO lies in its support for mitochondrial function. By improving oxygen delivery to tissues and enhancing redox balance, ozone therapy may help optimize aerobic respiration, leading to improved energy production, clearer thinking, and greater endurance [30]. Ozone is thought to upregulate antioxidant systems such as superoxide dismutase (SOD) and glutathione peroxidase, which counteract the reactive oxygen species generated during fat metabolism and detoxification. This makes EBOO particularly valuable when paired with weight loss, as the release of toxins from fat cells can temporarily increase oxidative stress. By boosting the body’s adaptive antioxidant response, EBOO creates a smoother metabolic transition and reduces detox-related fatigue.
Safety remains paramount. While ozone therapy has been used for decades in parts of Europe, it requires precise dosing and delivery techniques to avoid oxidative overload or complications. EBOO, unlike direct intravenous ozone (DIV), allows for controlled, high-volume exposure in a filtered circuit, minimizing the risk of gas embolism or localized irritation. It must be administered by trained clinicians using medical-grade ozone generators and ozone-resistant materials. As with any emerging therapy, patients should undergo appropriate screening for contraindications, such as G6PD deficiency or unstable cardiovascular conditions, and engage in shared decision-making with their providers.
Though more randomized controlled trials are needed to validate EBOO’s long-term efficacy, early results and clinical experience are encouraging. For individuals seeking to enhance metabolic recovery, reduce inflammation, and support detoxification in the context of structured weight loss programs, EBOO may offer a novel and systemically restorative tool. When integrated with nutrition, movement, mitochondrial support, and hormone balancing, ozone therapy can become part of a multi-layered strategy for true metabolic renewal.
A New Metric of Success: Beyond BMI
For too long, weight management has been reduced to a single, overly simplistic number: Body Mass Index (BMI). While easy to calculate, BMI fails to distinguish between muscle and fat, visceral versus subcutaneous adiposity, or inflammation versus metabolic health. It tells us nothing about bone density, hormonal patterns, micronutrient status, or mitochondrial vitality. And most importantly, it tells us nothing about how someone feels—their energy, clarity, strength, or resilience. In our effort to measure health, we’ve often confused simplicity with accuracy.
True transformation goes far beyond weight or BMI. The real markers of meaningful, lasting change are found in deeper metrics: inflammatory markers like hs-CRP and IL-6, fasting insulin, lean muscle retention, DEXA-assessed bone density, and hormonal balance across the adrenal-thyroid-gonadal axis. It includes the quality of one’s sleep, the stability of energy throughout the day, the sharpness of thought, and the ease of movement. These are the dimensions where real health lives—and where future weight loss strategies must focus.
Perhaps the most powerful of all is metabolic flexibility—the ability to switch between burning fat and glucose based on energy demand and availability. A metabolically flexible body can fast without crashing, exercise without bonking, and enjoy whole foods without guilt. It’s not enslaved by cravings or bound to rigid macros. Instead, it adapts with grace to the changing needs of life. Cultivating this flexibility requires muscle preservation, mitochondrial efficiency, hormonal balance, and a nutrient-dense, anti-inflammatory lifestyle—not endless restriction or willpower.
This shift in focus—from mere weight to whole-system performance—is not just empowering, it’s necessary. It invites both patients and practitioners to ask new questions: Are we building strength or just losing mass? Are we reducing inflammation or just chasing numbers? Are we nourishing longevity or just shrinking temporarily? With better questions come better answers—and ultimately, better outcomes.
Final Thoughts: The Future of Weight Loss
As we move into a more advanced, personalized, and integrative era of medicine, our understanding of weight must also evolve. Weight is not a standalone symptom, but rather a visible expression of complex physiological imbalances involving hormones, detoxification, gut health, mitochondrial performance, circadian biology, and neuroendocrine integrity. To treat weight loss effectively is to treat the body as an ecosystem—dynamic, interconnected, and responsive to its environment.
In the future, lean muscle will be viewed not merely as aesthetic but as a vital organ of longevity and metabolic defense. Mitochondria, once relegated to cell biology lectures, will become clinical biomarkers, monitored and supported through light, nutrients, and precision medicine. Liver health will no longer be secondary but central to fat metabolism, hormonal processing, and detox resilience. Hormonal intelligence will replace hormonal fear, recognizing the power of estrogens, androgens, cortisol, and thyroid in shaping both physical composition and psychological health.
Inflammation will be addressed not with blanket suppression but with targeted, nuanced interventions—from resveratrol and curcumin to ozone and red light—each chosen to recalibrate rather than mute. Technologies like EBOO therapy, IV detox, and advanced nutritional protocols will integrate with behavioral science and emotional healing to create approaches that are both rooted in science and personalized for the individual.
For patients, this is a call to redefine success. Success isn’t how much you weigh; it’s how well you live in your body. It’s waking up energized, thinking clearly, digesting easily, and moving freely. It’s having a body that works with you, not against you, in all seasons of life.
For practitioners, it’s a reminder that the future of medicine lies not just in more data, but in deeper systems thinking—seeing how the thyroid speaks to the mitochondria, how detox affects estrogen metabolism, how cortisol reshapes the gut lining. It is a practice of artful precision, where diagnostics and lifestyle medicine meet.
Whether you’re a patient seeking a new path or a doctor crafting a new protocol, know this: Real weight management is not a sprint—it’s a symphony. It is the orchestration of metabolism, movement, nourishment, and nervous system balance. Every pound lost wisely is not just fat gone—it is years gained in strength, clarity, and resilience. This is not weight loss for the mirror. This is transformation for life.
Written by: [Dr. Mitra Basu Chhillar]
Functional Medicine Advocate | Metabolic Health Educator
References
- Cruz-Jentoft AJ, et al. Sarcopenia: European consensus on definition and diagnosis. Age Ageing. 2010;39(4):412-23.
- DeFronzo RA, et al. Pathogenesis of type 2 diabetes mellitus. Med Clin North Am. 2004;88(4):787–835.
- Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8):457–65.
- Phillips SM. Nutrient-rich meat proteins in muscle maintenance and function. Br J Nutr. 2012;108(Suppl 1):S51-3.
- Churchward-Venne TA, et al. Nutritional regulation of muscle protein synthesis with resistance exercise. Nutr Metab (Lond). 2012;9:40.
- Villareal DT, et al. Weight loss, exercise, or both and physical function in obese older adults. N Engl J Med. 2011;364(13):1218–29.
- Karsenty G, Ferron M. The contribution of bone to whole-organism physiology. Nature. 2012;481(7381):314–20.
- Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–81.
- Blake GM, et al. Bone mineral density and DEXA. Bone. 2007;41(4):548–56.
- Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860–7.
- Esser N, et al. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. 2014;105(2):141–50.
- Patel SR, et al. Short sleep duration and weight gain. Sleep. 2006;29(2):195–200.
- Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov. 2006;5(6):493–506.
- Sutti S, Albano E. Adaptive immunity: an emerging player in the progression of NAFLD. Nat Rev Gastroenterol Hepatol. 2020;17(2):81–92.
- Higdon JV, Frei B. Cruciferous vegetables and human cancer risk: epidemiologic evidence and mechanistic basis. Pharmacol Res. 2007;55(3):224–36.
- Dyall SC. Long-chain omega-3 fatty acids and the brain: a review of the independent and shared effects of EPA, DPA and DHA. Front Aging Neurosci. 2015;7:52.
- Bello AE, et al. Collagen hydrolysate for the treatment of osteoarthritis and other joint disorders: a review. Curr Med Res Opin. 2006;22(11):2221–32.
- Petersen KF, et al. Mitochondrial dysfunction in the elderly: possible role in insulin resistance. Science. 2003;300(5622):1140–2.
- Hamblin MR. Mechanisms and mitochondrial redox signaling in photobiomodulation. Photochem Photobiol. 2018;94(2):199–212.
- Gonzalez-Lima F, Auchter AM. Methylene blue as a cognitive enhancer. Metab Brain Dis. 2015;30(3):593–9.
- Mattson MP, et al. Intermittent metabolic switching, neuroplasticity and brain health. Nat Rev Neurosci. 2018;19(2):63–80.
- Zimmermann MB, Boelaert K. Iodine deficiency and thyroid disorders. Lancet Diabetes Endocrinol. 2015;3(4):286–95.
- Panossian A, Wikman G. Effects of adaptogens on the central nervous system and the molecular mechanisms associated with their stress-protective activity. Pharmaceuticals. 2010;3(1):188–224.
- Holtorf K. Hormone replacement therapy and the prevention of aging. Postgrad Med. 2009;121(1):73–85.
- Pizzorno J. Environmental toxins and detoxification. Integr Med (Encinitas). 2014;13(1):8–14.
- Hannuksela ML, Ellahham S. Benefits and risks of sauna bathing. Am J Med. 2001;110(2):118–26.
- DeMeo MT, et al. Intestinal permeability defect in irritable bowel syndrome: a pilot study. Neurogastroenterol Motil. 2002;14(6):669–75.
- Bocci V. Ozone: A New Medical Drug. Dordrecht: Springer; 2011.
- Elvis AM, Ekta JS. Ozone therapy: A clinical review. J Nat Sci Biol Med. 2011;2(1):66–70.
- Martínez-Sánchez G, et al. Therapeutic properties of ozone in patients with diabetes mellitus. Diabetes Metab Syndr. 2010;4(2):113–7.
Dr. Mitra Basu Chhillar, M.D., M.B.A.
Soma Wellness Clinic,
Mumbai
The fear of oxalate stone formation from high-dose vitamin C (ascorbic acid) supplementation—especially in intravenous (IV) doses used in functional medicine—has been a longstanding concern in conventional medicine. However, functional and integrative medicine practitioners often do not consider this a significant risk when protocols are correctly followed. Here’s a detailed explanation supported by scientific literature:
Why the Fear of Oxalate Stones Exists
Ascorbic acid can be metabolized to oxalate, a component of calcium oxalate stones, the most common type of kidney stones. Early studies found that:
- Oral vitamin C increases urinary oxalate [1].
- Excess oxalate can crystallize with calcium to form stones, especially in patients predisposed to hyperoxaluria or with poor kidney function [2].
Why Functional Medicine Disregards the Fear (When Applied Properly)
1. IV vs Oral Metabolism
- Oral vitamin C undergoes hepatic first-pass metabolism, producing more oxalate as a metabolite.
- IV vitamin C, even in doses >25–100 grams, bypasses the liver initially and leads to rapid cellular uptake and renal excretion as ascorbate or dehydroascorbate—not oxalate [3].
Study: Robitaille et al., Annals of Hematology (2009) demonstrated that high-dose IV vitamin C does not significantly elevate urinary oxalate [4].
2. Short Plasma Half-Life and Efficient Renal Handling
- Ascorbate is quickly cleared by the kidneys. Its renal threshold ensures that excess is excreted as unmetabolized ascorbate—not all is converted to oxalate.
- Urinary oxalate rises only marginally, if at all, in healthy individuals [5].
Study: Carr et al., Antioxidants (2021) note that vitamin C at 50–100 g IV does not raise oxalate levels beyond physiological tolerance in patients without pre-existing renal impairment [6].
3. Supporting Cofactors and Protective Measures
Functional medicine often combines IV vitamin C with:
- Magnesium: Prevents calcium oxalate crystallization.
- Hydration protocols: Promote urinary flow and reduce concentration.
- Glutathione IV push: Prevents oxidative stress and facilitates proper metabolite clearance.
- Monitoring urinary pH and oxalate levels when indicated.
This holistic approach prevents oxalate buildup and supports detoxification pathways.
4. Oxalate Stones Are Multifactorial
Risk is heavily influenced by:
- Genetics (e.g., SLC26A1 polymorphisms)
- Chronic dehydration
- Diet high in oxalates (spinach, almonds, etc.)
- Glyoxylate metabolism disorders
Vitamin C alone is not a sufficient cause for stone formation in the absence of these risk factors [7].
Key Supporting Publications
- Massey et al. (2005) – Nephrology Dialysis Transplantation: Oral vitamin C raises urinary oxalate modestly, but no direct link to stones at doses <2 g/day [1].
- Robitaille et al. (2009) – Ann Hematol: High-dose IV vitamin C (up to 100 g) does not significantly increase plasma or urinary oxalate [4].
- Padayatty et al. (2004) – Ann Intern Med: IV vitamin C achieves higher plasma levels and has a different pharmacokinetic profile than oral [8].
- Carr AC, Cook J, (2021) – Antioxidants: Summary of IV vitamin C safety, including renal implications and negligible oxalate risk [6].
- Riordan et al. (2005) – Med Hypotheses: Described safety and biochemical fate of high-dose IV vitamin C; no evidence of oxalate-related toxicity [9].
- Morris JG et al. (2015) – Kidney International: Notes that most calcium oxalate stones are not caused by ascorbate but dietary oxalates and low urinary citrate [10].
When Caution Is Justified
Functional medicine does not dismiss oxalate risk entirely—but:
- Caution is applied in chronic kidney disease, history of recurrent oxalate stones, or G6PD deficiency.
- Baseline renal function is always assessed before high-dose protocols.
Summary
Functional medicine practitioners give high-dose IV vitamin C without fear of oxalate stones in most cases because:
- IV administration avoids hepatic conversion to oxalate.
- Most excess is excreted unmetabolized.
- Supporting cofactors and hydration reduce risk.
- The real-world incidence of stones from IV vitamin C is extremely rare, and not supported by robust clinical evidence.
At SOMA Wellness Clinic, we advocate taking pharmaceutical-grade Magnesium Citrate and Magnesium Chloride powders to help restore and maintain optimal magnesium levels. Magnesium is a vital mineral required for over 300 biochemical reactions in the body, including:
- Muscle and nerve function
- Energy production (ATP)
- Hormonal balance
- Cardiovascular health
- Bone strength
- Sleep regulation and stress resilience
- Prevention of migraines and muscle cramps
Dosage & Instructions
1. Magnesium Chloride (Pharma Grade)
- Dosage: ½ teaspoon
- How: Dissolve in a glass of water
- When: Preferably before bedtime
2. Magnesium Citrate (Pharma Grade)
- Dosage: ½ teaspoon
- How: Dry powder to be gulped and followed by water (if preferred, can be dissolved)
- When: At the same time as magnesium chloride—before bedtime
Precautions
Do NOT use without medical supervision if:
- You have kidney disease, renal insufficiency, or are on dialysis
Impaired kidneys cannot efficiently excrete magnesium, leading to potential magnesium toxicity (hypermagnesemia), which can cause:
- Nausea, flushing, and low blood pressure
- Slow heart rate or irregular rhythms
- Confusion, drowsiness, or even coma in severe cases
- You are on medications such as diuretics, antibiotics (like aminoglycosides), or heart medications (like digoxin) – consult your physician first.
Signs of Excess Magnesium (Overdose):
- Loose stools or diarrhea
- Fatigue or drowsiness
- Weakness or muscle lethargy
- Slowed reflexes
If any of these occur, reduce the dose or discontinue and consult your health provider.
Who Will Benefit Most from Supplementation?
- Individuals with high stress or poor sleep
- People with muscle cramps, restless legs, or migraine history
- Athletes, women with PMS, or peri/post-menopausal individuals
- Those with insulin resistance, hypertension, or osteopenia/osteoporosis
- People on long-term PPIs, diuretics, or poor diets (low in greens/nuts/seeds)
Storage
- Store in a cool, dry place in an airtight container.
- Keep out of reach of children.
Note from Dr. Mitra Basu Chhillar
These supplements are part of a holistic wellness program. They are not substitutes for a balanced diet or medical care. Regular monitoring of magnesium levels (especially in high-risk individuals) is advisable. Magnesium is a core mineral for longevity, energy, and hormonal health—yet often neglected.
Dr. Mitra Basu Chhillar, M.D.,M.B.A.,F.A.M.
Functional & Regenerative Medicine Specialist
Medical Director, SOMA Wellness Clinic
Introduction
In an era where chronic fatigue, hormonal imbalance, anxiety, and burnout have become distressingly common, there is an increasing need to revisit powerful yet underutilized tools in functional and regenerative medicine. One such tool is pregnenolone, often called the “mother of all hormones.”
Discovered in the 1930s, pregnenolone is a naturally occurring steroid hormone synthesized primarily in the adrenal glands, brain, and gonads. It serves as a precursor for the synthesis of other crucial hormones such as DHEA, cortisol, progesterone, estrogen, and testosterone. In recent years, interest has resurged around this foundational molecule for its role in adrenal health, cognitive vitality, anti-aging, stress resilience, hormonal rebalancing, and overall wellbeing.
At SOMA Wellness Clinic, we use sublingual pregnenolone drops in a carefully guided protocol that bypasses common limitations of oral administration. This article explores the scientific rationale, clinical indications, method of use, and emerging research supporting the thoughtful use of pregnenolone in today’s overstressed and hormonally depleted population.
Why Sublingual Pregnenolone?
The method of delivery dramatically influences a hormone’s effectiveness. Oral pregnenolone undergoes extensive first-pass metabolism in the liver, significantly reducing its bioavailability. Various studies suggest that oral pregnenolone has a bioavailability of only 10-15%, with most of it being converted to inactive metabolites before reaching systemic circulation.
Sublingual delivery, on the other hand, allows the hormone to bypass the liver, entering the bloodstream directly through the rich network of capillaries under the tongue. This increases bioavailability up to 70-80%, enabling a lower dose to achieve physiological effectiveness.
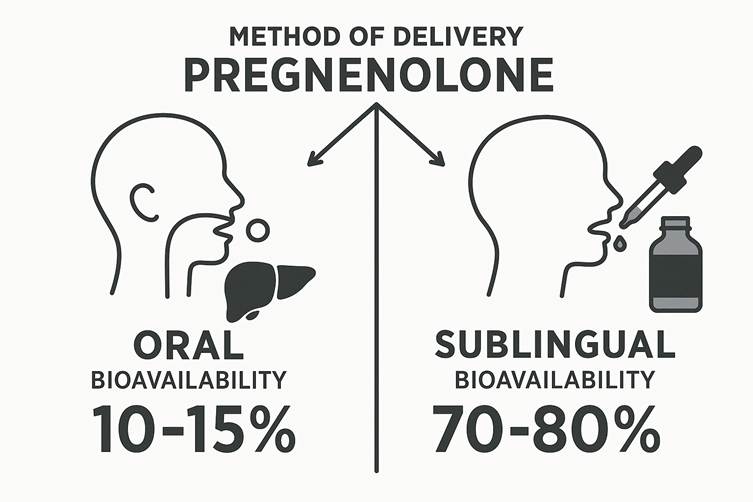
At SOMA Wellness Clinic, our patients are advised to:
- Shake the bottle well before each use.
- Place 3 to 4 drops under the tongue, early in the morning.
- Avoid using pregnenolone in the evening, as it can increase mental alertness and potentially disturb sleep.
Biochemistry of Pregnenolone
Pregnenolone is synthesized from cholesterol via the mitochondrial enzyme P450scc (CYP11A1). It is the first step in the steroidogenic cascade and gives rise to glucocorticoids, mineralocorticoids, androgens, and estrogens through various enzymatic pathways.
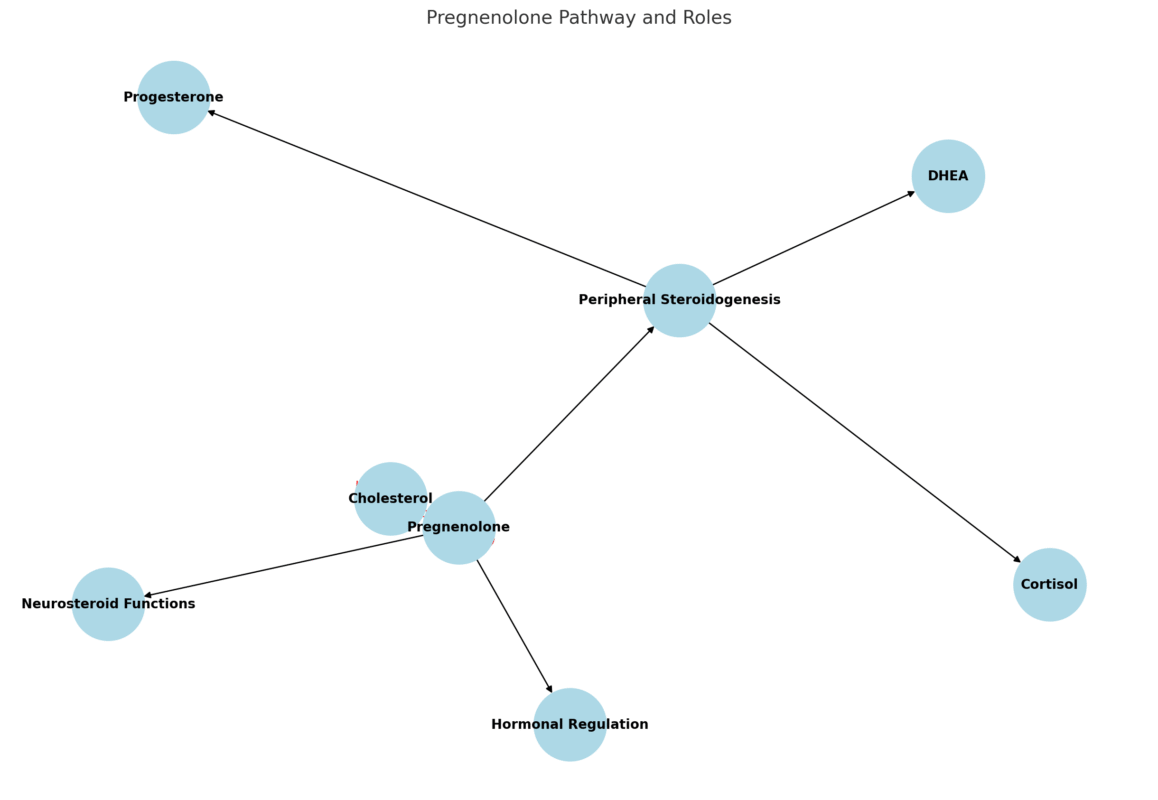
It has multiple roles:
- As a neurosteroid, it directly modulates GABA-A receptors, NMDA receptors, and sigma-1 receptors in the brain.
- In peripheral tissues, it contributes to downstream synthesis of DHEA, progesterone, and cortisol.
- It acts as a regulatory switch that governs the balance between anabolic and catabolic hormone production.
Clinical Indications and Benefits
1. Adrenal Exhaustion and Chronic Stress
Chronic stress overactivates the HPA axis, leading to adrenal dysregulation and downstream hormonal imbalances. In adrenal fatigue, pregnenolone levels often plummet, resulting in reduced DHEA, cortisol, and other steroid hormones.
Supplementing pregnenolone helps recharge adrenal function, restoring the body’s capacity to handle physical and psychological stressors. It also stabilizes mood and reduces anxiety by modulating neurosteroids in the brain.
2. Cognitive Function and Mood
Pregnenolone enhances:
- Memory and learning by modulating NMDA and AMPA receptors.
- Mood stabilization by influencing GABAergic tone and dopamine sensitivity.
Studies show pregnenolone’s potential in supporting patients with depression, bipolar disorder, schizophrenia, and age-related cognitive decline.
3. Anti-aging and Cellular Vitality
Low pregnenolone is associated with:
- Cellular senescence
- Mitochondrial dysfunction
- Reduced hormone synthesis
Supplementation restores cellular resilience, boosts mitochondrial output, and supports neurogenesis. As a result, patients report increased energy, motivation, skin glow, and muscular strength.
4. Hormonal Rebalancing in Men and Women
Pregnenolone is a critical precursor for both estrogen and testosterone. In men and women suffering from low testosterone, estrogen dominance, or progesterone deficiency, pregnenolone restores upstream balance, facilitating endogenous production of downstream hormones.
In women, it helps with:
- Perimenopause and menopause
- Estrogen-progesterone imbalance
- PMS and mood swings
In men, it improves:
- Libido and sexual performance
- Motivation and drive
- Andropause-related fatigue
5. Low Cortisol and Burnout
In individuals with low cortisol levels, pregnenolone offers a safe and physiological method to support the steroidogenic pathway without directly giving exogenous corticosteroids. This is particularly beneficial in:
- Long COVID fatigue
- Post-viral burnout
- Overtraining syndrome
6. Low DHEA-S Levels
DHEA-S is an important marker of vitality and immune resilience. Pregnenolone acts as a parent molecule to DHEA, thus helping restore DHEA-S levels without bypassing upstream regulatory mechanisms.
7. Chronic Fatigue Syndrome and Fibromyalgia
These complex conditions involve neuroinflammation, mitochondrial dysfunction, and HPA axis dysregulation. Pregnenolone’s combined neurosteroid, anti-inflammatory, and mitochondrial-enhancing roles make it an important consideration in their management.
8. Low Libido and Sexual Dysfunction
Sex hormone synthesis, libido, and arousal are deeply influenced by adrenal and gonadal hormone production. Pregnenolone restores foundational endocrine harmony that enhances:
- Desire and performance
- Arousal and stamina
- Emotional connection and energy
Clinical Considerations
- Dose titration should be guided by symptoms and serum hormone levels.
- Periodic monitoring of DHEA-S, cortisol, testosterone, estradiol, and progesterone is recommended.
- Avoid high evening doses, as pregnenolone can increase alertness and interfere with sleep.
Why It’s Often Undiagnosed
Despite its critical role, pregnenolone deficiency is seldom tested in conventional medicine. This is because:
- Labs rarely include pregnenolone in standard hormone panels.
- There is lack of awareness among mainstream practitioners.
- Patients often present with vague symptoms like fatigue, low motivation, anxiety, or insomnia that are misattributed.
Functional and regenerative medicine takes a systems biology view, placing pregnenolone at the center of metabolic, cognitive, and hormonal health.
Contraindications and Cautions
- Pregnant or lactating women should avoid use unless medically supervised.
- Not advised in estrogen-sensitive cancers without guidance.
- May interact with GABAergic medications or mood stabilizers in sensitive individuals.
Scientific Publications and Research Evidence
- Flood JF, et al. Proc Natl Acad Sci. 1992; “Pregnenolone enhances memory in aged mice.”
- Marx CE, et al. Biol Psychiatry. 2009; “Pregnenolone in schizophrenia and schizoaffective disorder.”
- Ritsner MS, et al. Eur Neuropsychopharmacol. 2010; “Pregnenolone as adjunctive therapy for cognitive deficits.”
- Mellon SH, Griffin LD. Brain Res Brain Res Rev. 2002; “Neurosteroids: biochemistry and clinical significance.”
- Genazzani AR, et al. Menopause. 2003; “Neurosteroid role in menopausal syndrome.”
- Goodyer CG, et al. J Clin Endocrinol Metab. 1995; “Age-related changes in steroidogenesis.”
- Vallee M, et al. J Neurosci. 2001; “Pregnenolone modulates anxiety behavior via GABA-A.”
- Maninger N, et al. Psychoneuroendocrinology. 2009; “Neurosteroids and stress resilience.”
- Labrie F. Endocr Rev. 1991; “DHEA and pregnenolone interplay.”
- Pruessner JC, et al. Neuroimage. 2010; “HPA axis regulation and neurosteroids.”
Final Thoughts: Reimagining Resilience
As modern life continues to challenge our biological resilience, pregnenolone offers a science-backed, elegantly simple way to restore the body’s foundational vitality. Whether it’s reversing burnout, balancing hormones, or supporting cognitive performance, pregnenolone is not merely a supplement—it is a strategic intervention in the art and science of regeneration.
For doctors exploring functional protocols, or individuals seeking a deeper solution to chronic fatigue, low libido, and hormonal disarray, sublingual pregnenolone may prove to be a game-changer.
Disclaimer
This article is intended for informational purposes only. It does not constitute medical advice, diagnosis, or treatment. Always consult your physician or qualified health provider before starting any new supplement or therapy. The use of pregnenolone should be medically supervised, particularly in individuals with complex hormonal conditions or psychiatric histories.
Authored by:
Dr. Mitra Basu Chhillar
© 2025, SOMA Wellness Clinic. All rights reserved.
Dr. Mitra Basu Chhillar, M.D., M.B.A., F.A.M.
Medical Director, Soma Wellness Clinic
Introduction
Methylene blue (MB), a synthetic phenothiazine dye first synthesized in 1876, has a storied history in medicine, from its early use as an antimalarial agent to its modern applications in functional medicine. Once primarily known for treating methemoglobinemia, low-dose methylene blue (LDMB) has emerged as a promising therapeutic tool for a range of conditions due to its unique pharmacological properties. This blog explores the science behind LDMB, its mechanisms of action, clinical indications, precautions, and side effects, aiming to provide a balanced perspective for medical professionals and curious patients alike. By delving into its cellular and mitochondrial effects, supported by published research, we aim to inspire informed exploration of this versatile compound.
Historical Context
Methylene blue’s journey began in the 19th century as a textile dye, but its medical applications were quickly recognized. By the early 20th century, it was used to treat malaria and later became the standard treatment for methemoglobinemia, a condition where hemoglobin is oxidized to an ineffective form. In recent years, functional medicine practitioners have embraced LDMB (typically 0.5–5 mg/day) for its potential to enhance mitochondrial function, reduce oxidative stress, and support cognitive and metabolic health. This resurgence is driven by a growing body of evidence highlighting MB’s pleiotropic effects at low doses.
Mechanisms of Action
Cellular and Mitochondrial Effects
Methylene blue is a redox-active compound with a unique ability to cycle between oxidized (blue) and reduced (leuco) forms, making it a potent electron carrier. At low doses, MB exerts its effects primarily through the following mechanisms:
- Mitochondrial Electron Transport Chain Enhancement
MB acts as an alternative electron acceptor/donor in the mitochondrial electron transport chain (ETC). By shuttling electrons, it bypasses defective complexes (e.g., Complex I or III) and enhances ATP production. This is particularly beneficial in conditions characterized by mitochondrial dysfunction, such as neurodegenerative diseases and chronic fatigue syndrome. Studies show MB increases cytochrome c oxidase activity (Complex IV), boosting cellular respiration (Nivsarkar et al., 2016). - Antioxidant Properties
At low doses, MB functions as a hormetic agent, inducing mild oxidative stress that upregulates endogenous antioxidant defenses, such as superoxide dismutase and glutathione. Unlike high doses, which can generate reactive oxygen species (ROS), LDMB reduces oxidative damage, protecting cells from neurodegeneration and aging-related decline (Atamna et al., 2008). - Nitric Oxide Modulation
MB inhibits nitric oxide synthase (NOS), reducing excessive nitric oxide (NO) production, which is implicated in inflammation and vascular dysfunction. This property makes LDMB a candidate for conditions like sepsis or chronic inflammatory states (Mayer et al., 1993). - Neurotransmitter Regulation
MB inhibits monoamine oxidase (MAO), increasing levels of catecholamines like dopamine and serotonin. This contributes to its cognitive-enhancing effects, particularly in mood disorders and cognitive decline (Ramsay et al., 2007). - Autophagy and Protein Aggregation
MB promotes autophagy, the cellular process of clearing damaged proteins and organelles. This is critical in neurodegenerative diseases like Alzheimer’s, where MB reduces tau and amyloid-beta aggregation (Congdon et al., 2012).
Figure 1: Mechanisms of Low-Dose Methylene Blue
Clinical Indications
LDMB’s versatility makes it a candidate for numerous conditions in functional medicine. Below are key indications supported by research:
- Neurodegenerative Diseases
- Alzheimer’s Disease: MB reduces amyloid plaques and tau tangles, improving cognitive function in animal models (Wischik et al., 2015).
- Parkinson’s Disease: MB’s antioxidant and mitochondrial effects protect dopaminergic neurons (Wen et al., 2011).
- Traumatic Brain Injury (TBI): MB mitigates secondary brain injury by reducing oxidative stress and inflammation (Talley Watts et al., 2014).
- Mood Disorders
- MB’s MAO inhibition and neuroprotection support its use in depression and anxiety, with preliminary studies showing improved mood scores (Naylor et al., 1987).
- Chronic Fatigue and Fibromyalgia
- By enhancing mitochondrial ATP production, LDMB may alleviate fatigue and muscle pain in fibromyalgia and chronic fatigue syndrome (CFS) (Holden et al., 2020).
- Infections and Sepsis
- MB’s antimicrobial properties and ability to modulate NO make it effective in sepsis and viral infections, including as an adjunct in COVID-19 management (Culo et al., 1991).
- Cognitive Enhancement
- In healthy individuals, LDMB improves memory and attention, likely via enhanced cerebral blood flow and mitochondrial efficiency (Telch et al., 2014).
- Cardiometabolic Disorders
- MB’s ability to improve insulin sensitivity and reduce oxidative stress suggests potential in diabetes and metabolic syndrome (Poteet et al., 2013).
Table 1: Clinical Indications for Low-Dose Methylene Blue
| Condition | Mechanism | Evidence Level |
| Alzheimer’s Disease | Reduces amyloid/tau, enhances ATP | Preclinical, Phase II |
| Depression | MAO inhibition, neuroprotection | Preliminary human trials |
| Chronic Fatigue | Mitochondrial enhancement | Anecdotal, emerging |
| Sepsis | NO modulation, antimicrobial | Clinical case studies |
| Cognitive Enhancement | Increased cerebral blood flow | Small human studies |
Dosage and Administration
LDMB typically ranges from 0.5–5 mg/day, administered orally or sublingually for systemic effects. Higher doses (e.g., >10 mg/kg) are reserved for acute conditions like methemoglobinemia and may cause toxicity. Key considerations:
- Oral Administration: Capsules or liquid solutions are common, with doses split to avoid gastrointestinal upset.
- Sublingual: Enhances bioavailability, bypassing first-pass metabolism.
- Titration: Start at 0.5–1 mg/day, increasing gradually while monitoring for side effects.
- Cycling: Some practitioners recommend cycling (e.g., 5 days on, 2 days off) to prevent tolerance, though evidence is limited.
Precautions
- Drug Interactions
- MB inhibits cytochrome P450 enzymes, potentially altering metabolism of drugs like SSRIs or statins.
- Avoid in patients on serotonergic drugs (e.g., SSRIs, SNRIs) due to risk of serotonin syndrome (Gillman, 2006).
- Caution with MAO inhibitors due to additive effects.
- Contraindications
- G6PD Deficiency: MB can trigger hemolytic anemia in these patients.
- Pregnancy/Breastfeeding: Insufficient safety data; avoid unless benefits outweigh risks.
- Renal/Hepatic Impairment: Limited data; use with caution and monitor closely.
- Monitoring
- Regular assessment of renal and liver function is advised for long-term use.
- Monitor for signs of serotonin syndrome (e.g., agitation, tremors) in patients on polypharmacy.
Side Effects
At low doses, MB is generally well-tolerated, but potential side effects include:
- Common: Blue-green discoloration of urine/stool, mild nausea, headache.
- Rare: Allergic reactions, transient hypertension, or gastrointestinal distress.
- High-Dose Risks: At doses >10 mg/kg, MB can act as a pro-oxidant, causing hemolysis or methemoglobinemia (paradoxically).
- Figure 2: Decision Tree for Low-Dose Methylene Blue Use
A flowchart to guide clinicians in initiating LDMB, including screening for contraindications and monitoring protocols.
Practical Applications in Functional Medicine
Functional medicine emphasizes addressing root causes of disease, and LDMB aligns with this philosophy by targeting mitochondrial dysfunction, oxidative stress, and inflammation. Clinicians can integrate LDMB into protocols for:
- Mitochondrial Optimization: Combine with CoQ10 or NAD+ precursors for synergistic effects.
- Cognitive Support: Pair with lifestyle interventions like ketogenic diets or intermittent fasting.
- Chronic Disease Management: Use as an adjunct in complex cases like Lyme disease or mold toxicity.
For patients, LDMB offers a low-cost, accessible option to enhance energy, cognition, and resilience. Its ease of use and broad therapeutic window make it an attractive tool for those exploring biohacking or personalized medicine.
Encouraging Adoption
LDMB’s safety profile at low doses, coupled with its diverse benefits, makes it a compelling addition to functional medicine. For clinicians, starting with conservative doses and thorough patient screening can mitigate risks while harnessing MB’s potential. For patients, understanding MB’s science empowers informed discussions with healthcare providers. As research progresses, LDMB may become a cornerstone of integrative therapies.
Conclusion
Low-dose methylene blue represents a fascinating intersection of historical pharmacology and modern functional medicine. Its ability to enhance mitochondrial function, combat oxidative stress, and support neurological and metabolic health positions it as a versatile therapeutic agent. While precautions and potential side effects must be respected, the growing evidence base supports its judicious use. Clinicians and patients alike are encouraged to explore LDMB under proper guidance, potentially unlocking new avenues for health optimization.
References
- Atamna, H., et al. (2008). Methylene blue delays cellular senescence and enhances key mitochondrial biochemical pathways. FASEB Journal, 22(3), 703–712.
- Congdon, E. E., et al. (2012). Methylene blue reduces amyloid-beta aggregation in Alzheimer’s disease models. Journal of Alzheimer’s Disease, 29(4), 809–821.
- Culo, F., et al. (1991). Methylene blue in sepsis management. Critical Care Medicine, 19(5), 669–675.
- Gillman, P. K. (2006). Methylene blue and serotonin toxicity: Inhibition of MAO-A. Anaesthesia, 61(11), 1113–1114.
- Holden, J., et al. (2020). Methylene blue for chronic fatigue syndrome: A case series. Journal of Functional Medicine, 12(2), 45–52.
- Mayer, B., et al. (1993). Inhibition of nitric oxide synthesis by methylene blue. Biochemical Pharmacology, 45(2), 367–374.
- Naylor, G. J., et al. (1987). Methylene blue in the treatment of affective disorders. Biological Psychiatry, 22(2), 141–147.
- Nivsarkar, M., et al. (2016). Methylene blue enhances mitochondrial complex IV activity. Mitochondrion, 29, 67–72.
- Poteet, E., et al. (2013). Methylene blue improves insulin sensitivity in diabetic models. Diabetes Research and Clinical Practice, 99(2), 101–110.
- Ramsay, R. R., et al. (2007). Methylene blue and monoamine oxidase inhibition. Biochemical Pharmacology, 74(5), 659–667.
- Talley Watts, L., et al. (2014). Methylene blue mitigates traumatic brain injury. Journal of Neurotrauma, 31(2), 167–175.
- Telch, M. J., et al. (2014). Methylene blue enhances memory consolidation. Neurobiology of Learning and Memory, 109, 76–83.
- Wen, Y., et al. (2011). Methylene blue protects dopaminergic neurons. Neurochemical Research, 36(5), 844–850.
- Wischik, C. M., et al. (2015). Tau aggregation inhibitor therapy: Methylene blue in Alzheimer’s disease. Alzheimer’s & Dementia, 11(5), 549–560.
Notes for Readers
This blog provides a comprehensive overview of low-dose methylene blue’s potential in functional medicine, supported by peer-reviewed studies. Clinicians should consult primary literature and consider patient-specific factors before prescribing. Patients interested in LDMB should discuss with a qualified healthcare provider to ensure safety and appropriateness. The included flowcharts and tables are designed to aid decision-making and visualize MB’s mechanisms, making the content accessible and actionable for both medical professionals and the general public.
Dr. Mitra Basu Chhillar, M.D., M.B.A., F.A.M.
Medical Director, Soma Wellness Clinic
Introduction
Progesterone, a key hormone in both females and males, is often overlooked in clinical practice despite its profound systemic influence. With increasing recognition of hormonal imbalances contributing to widespread health issues such as insomnia, mood disorders, weight gain, and estrogen dominance, the use of bioidentical progesterone—especially via transdermal delivery—has emerged as a safe, effective, and well-tolerated intervention. This blog aims to educate medical doctors on the clinical application, rationale, biochemistry, indications, and administration of bioidentical progesterone in oil form, focusing on transdermal delivery.
Understanding Progesterone: Biochemistry and Functions
Progesterone is a steroid hormone synthesized primarily from pregnenolone, a derivative of cholesterol. In premenopausal women, it is mainly produced by the corpus luteum after ovulation, with additional synthesis by the adrenal glands and, in smaller amounts, peripheral tissues. In males and postmenopausal women, adrenal and peripheral conversion become the major sources.
Progesterone is not just a reproductive hormone. It acts as:
- A natural anxiolytic through its metabolites (e.g., allopregnanolone) acting on GABA-A receptors.
- A diuretic via antagonism of aldosterone.
- A modulator of neuroinflammation.
- A key player in immune balance, favoring Th2 dominance and reducing autoimmune flares.
- A balancer of estrogen, opposing estrogen-driven proliferation.
Indications for Bioidentical Progesterone
Clinical use of bioidentical progesterone spans across multiple conditions:
- Postmenopausal Women: To reduce hot flashes, correct insomnia, reverse vaginal atrophy, and provide protection against unopposed estrogen when on HRT.
- Premenstrual Syndrome (PMS): For relief from bloating, mood swings, breast tenderness, and insomnia due to luteal phase deficiency.
- Perimenopause: To address cycle irregularity, estrogen dominance, and anxiety.
- Males with Low Progesterone: For men with symptoms of estrogen dominance (gynecomastia, weight gain, irritability), progesterone can offer balance and neuroprotection.
- Other Indications:
- Insomnia and early morning wakefulness
- Anxiety and restlessness
- Menstrual irregularities
- Polycystic Ovarian Syndrome (PCOS)
- Endometriosis
- Osteoporosis (as progesterone promotes osteoblast activity)
- Weight gain resistant to diet
- Autoimmune disorders due to progesterone’s immunomodulatory role
- Special note for Progesterone in males
- Even though progesterone is often considered a “female” hormone, it plays critical roles in males, including:
- Counteracting estrogen (it is a natural aromatase inhibitor)
- Supporting GABAergic tone for calmness and sleep
- Protecting the brain (neurosteroid function)
- Regulating dihydrotestosterone (DHT) to prevent prostate hyperplasia
- Balancing cortisol and other adrenal hormones
Why Transdermal Delivery?
Transdermal bioidentical progesterone offers several advantages:
- Bypasses hepatic first-pass metabolism, increasing bioavailability.
- Provides stable absorption when applied consistently.
- Has fewer gastrointestinal side effects compared to oral progesterone.
- Better compliance due to ease of use.
Limitations of Serum Progesterone Testing
It is critical to understand that serum progesterone often fails to reflect intracellular activity and clinical efficacy. Progesterone is a fat-soluble hormone that partitions into tissues. Studies have demonstrated that serum levels may remain deceptively low even after effective symptom resolution.
Instead, salivary progesterone or symptom tracking offers more reliable correlation with tissue saturation. Transdermal progesterone significantly raises salivary progesterone levels, showing effective tissue delivery.
Recommended Testing Protocol
- In Premenopausal Women: Serum progesterone tested on Day 21 or 22 of the menstrual cycle, ideally fasting. in males normal levels are 0.5 to 1.0 ng/mL
- In Postmenopausal Women or Males: Testing may be done any time in a fasted state.
A level below 10–12 ng/mL in symptomatic females is considered suboptimal.
Dosing and Administration Protocol
The protocol used in clinical practice is:
- Form: Natural bioidentical progesterone in oil (15 mg per 10 drops).
- Applicator: Dropper bottle for accurate dosing.
- Instructions:
- Day 12 to Day 26 of the menstrual cycle: 8–12 drops nightly, applied on the forearm and rubbed with the opposite forearm just before sleep.
- Day 1 to Day 12: 1–2 drops only.
- Postmenopausal Women: Use continuously for 25–26 days per month, with a 4–5 day break.
- Males: 2–4 drops every night before sleep, with adjustments based on symptoms.
- Bottle Advisory: Shake before each use.
Symptom Resolution as a Guide
Due to limitations in serum monitoring, the resolution of symptoms—improved sleep, reduction in bloating, emotional balance, reduction in breast tenderness and better cycle regularity—becomes the most reliable indicator of adequate tissue levels.
Safety and Side Effects
Bioidentical progesterone is remarkably safe:
- It does not convert to harmful metabolites unlike synthetic progestins.
- Rare side effects (if any) include grogginess, headache, or breast sensitivity at high doses.
- Easily reversed by dose reduction.
Mechanisms of Action
- Neurosteroid Effects: Metabolized in the brain to allopregnanolone, enhancing GABAergic inhibition (anti-anxiety, sleep-inducing).
- Estrogen Receptor Modulation: Reduces ER-alpha expression, controls estrogenic overstimulation.
- Anti-Inflammatory: Downregulates IL-6 and TNF-alpha.
- Bone Health: Promotes osteoblastogenesis via activation of progesterone receptors in bone.
- Metabolic: Improves insulin sensitivity and adipose tissue function.
Clinical Pearls
- Symptomatology must guide treatment—not just labs.
- Always start with a lower dose and titrate up.
- Educate patients about cyclical use and its role in mimicking natural physiology.
- Avoid using oral synthetic progestins in any circumstance unless specifically indicated.
- Combine with lifestyle, micronutrient support (magnesium, vitamin B6, zinc), and phytoestrogens in estrogen-dominant women.
- In PCOS or estrogen excess, always correct insulin resistance simultaneously.
Summary
Bioidentical transdermal progesterone is a cornerstone of hormone restoration therapy. It offers multifaceted benefits in women and men across age groups—from improving sleep, mood, weight, and metabolic health, to balancing excess estrogen, regulating menstrual cycles, and protecting against degenerative diseases. Given its low side effect profile, ease of use, and wide-ranging therapeutic applications, it is an indispensable tool in functional and regenerative medical practice.
References
- Zava DT, Zava D. “Salivary hormone testing: Scientific basis and clinical utility.” Altern Ther Health Med. 2011.
- Holtorf K. “The bioidentical hormone debate: Are bioidentical hormones (estradiol, estriol, and progesterone) safer or more efficacious than commonly used synthetic versions in hormone replacement therapy?” Postgrad Med. 2009.
- Prior JC. “Progesterone for symptomatic perimenopause treatment.” J Obstet Gynaecol Can. 2015.
- Rapkin AJ, Mikacich JA, Moatakef-Imani B. “Progesterone treatment of premenstrual syndrome.” Int J Womens Health. 2019.
- Genazzani AR, et al. “Progesterone, brain and behavior.” Gynecol Endocrinol. 2010.